COLUMBA: Phase 3, randomized, open-label, non-inferiority, multicenter trial comparing DARZALEX FASPRO® monotherapy vs DARZALEX® monotherapy in 522 adult patients with relapsed or refractory multiple myeloma1,2


ECOG PS=Eastern Cooperative Oncology Group Performance Status; IV=intravenous; PD=progressive disease; QW=once weekly; Q2W=every 2 weeks; Q4W=every 4 weeks; sub-Q=subcutaneous.
*Including a proteasome inhibitor (PI) and an immunomodulatory agent, or double refractory to a PI and an immunomodulatory agent.1
Patient characteristics
Baseline demographics and disease characteristics were similar between the 2 treatment groups.2
- The median age was 67 years (range: 33–92 years). The median weight was 73 kg (range: 29–138 kg). Patients had received a median of 4 prior lines of therapy; 51% had a prior autologous stem cell transplant (ASCT) and 100% had received both a PI and an immunomodulatory agent1
Non-inferiority co-primary endpoints
Overall response rate† (ORR) and a maximum trough (Ctrough) concentration of daratumumab measured on Day 1 of Cycle 3.1
Selected secondary endpoints
Percentage of patients with systemic administration-related reactions‡ (ARRs), very good partial response (VGPR) or better rate, complete response (CR) or better rate, and progression-free survival (PFS).2
†According to International Myeloma Working Group (IMWG) response criteria.1
‡In clinical trials of DARZALEX FASPRO® and DARZALEX®, and in the Prescribing Information for DARZALEX®, the terms “infusion reactions” and “infusion-related reactions” were used instead of “systemic administration-related reactions.”
DARZALEX FASPRO® demonstrated efficacy consistent with DARZALEX®1
In COLUMBA, the co-primary endpoint was achieved:
DARZALEX FASPRO® was non-inferior to DARZALEX® regarding ORR.1
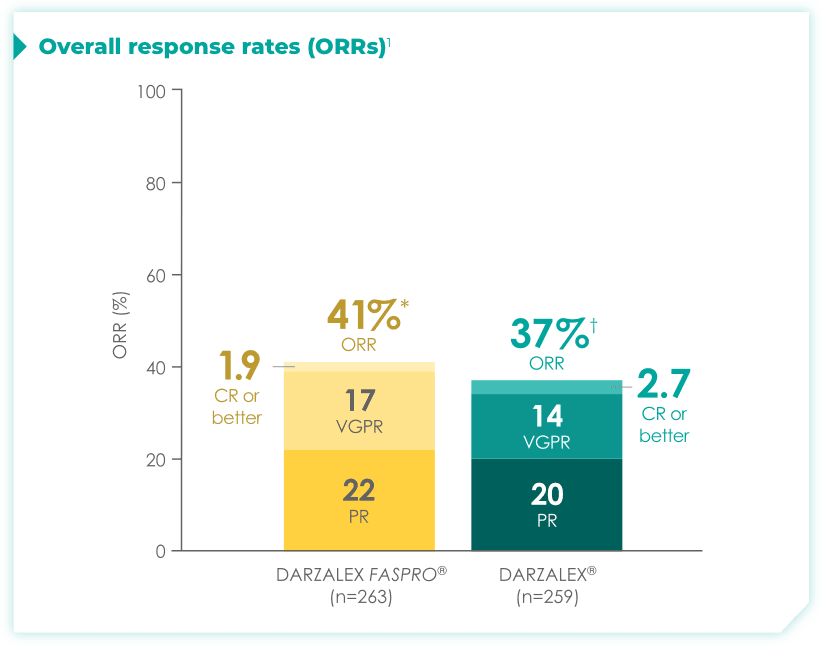

*DARZALEX FASPRO® (95% CI: 35, 47).
†DARZALEX® (95% CI: 31, 43).
The co-primary endpoint of remaining therapeutic drug concentration (maximum Ctrough) of DARZALEX FASPRO® was also non-inferior to DARZALEX®1
CI=confidence interval; CR=complete response; Ctrough=trough concentration; IV=intravenous; ORR=overall response rate; PR=partial response; sub-Q=subcutaneous; VGPR=very good partial response.
Fewer systemic ARRs1
Nearly 3x reduction in systemic administration-related reactions* (ARRs) with DARZALEX FASPRO® vs DARZALEX® were observed in the COLUMBA trial.1
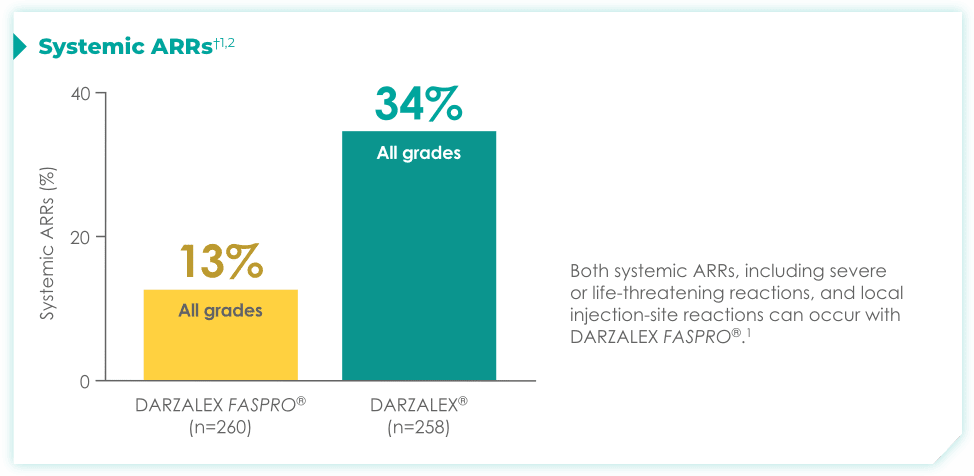

Most common systemic ARRs†1,2
Most systemic ARRs were Grade 1 or 2 and occurred with the first injection.
The most common systemic ARRs (DARZALEX FASPRO® vs DARZALEX®) were chills (5% vs 12%), pyrexia (5% vs 3%), and dyspnea (1% vs 7%).
Grade 3 systemic ARRs occurred in 2% of patients using DARZALEX FASPRO® vs 5% of patients on DARZALEX®.
No Grade 4 systemic ARRs were reported.
ARR=administration-related reaction; IV=intravenous; sub-Q=subcutaneous.
*In clinical trials of DARZALEX FASPRO® and DARZALEX®, and the Prescribing Information for DARZALEX®, the terms “infusion reactions” and “infusion-related reactions” were used instead of “systemic administration-related reactions.”
†Systemic ARRs causing severe reactions included hypoxia, dyspnea, hypertension, tachycardia, and ocular adverse reactions, including choroidal effusion, acute myopia, and acute angle closure glaucoma. Other signs and symptoms of systemic ARRs may include respiratory symptoms, such as bronchospasm, nasal congestion, cough, throat irritation, allergic rhinitis, and wheezing, as well as anaphylactic reaction, pyrexia, chest pain, pruritus, chills, vomiting, nausea, hypotension, and blurred vision.1
In a pooled safety population of 898 patients, the rate of systemic ARRs was 9% for DARZALEX FASPRO®1
- The median time to onset of systemic ARRs following an injection of DARZALEX FASPRO® was 3.2 hours (range: 4 minutes to 3.5 days). The majority of systemic ARRs occurred on the day of treatment. Delayed systemic ARRs, those occurring after the day of administration, have occurred in 1% of patients
- The incidence of any grade systemic ARRs was 8% with the first injection of DARZALEX FASPRO® at Week 1, 0.3% with the second injection at Week 2, and cumulatively 1% with subsequent injections
- Severe reactions included hypoxia, dyspnea, hypertension, tachycardia, and ocular adverse reactions, including choroidal effusion, acute myopia, and acute angle closure glaucoma. Other signs and symptoms of systemic administration-related reactions may include respiratory symptoms, such as bronchospasm, nasal congestion, cough, throat irritation, allergic rhinitis, and wheezing, as well as anaphylactic reaction, pyrexia, chest pain, pruritus, chills, vomiting, nausea, hypotension, and blurred vision
- Ocular adverse reactions, including acute myopia and narrowing of the anterior chamber angle due to ciliochoroidal effusions with potential for increased intraocular pressure or glaucoma, have occurred with daratumumab-containing products. If ocular symptoms occur, interrupt DARZALEX FASPRO® and seek immediate ophthalmologic evaluation prior to restarting DARZALEX FASPRO®
Local reactions1
- In this pooled safety population, injection-site reactions occurred in 8% of patients, including Grade 2 reactions in 0.7%. The most frequent (>1%) injection-site reaction was injection-site erythema
- These local reactions occurred a median of 5 minutes (range: 0 minutes to 6.5 days) after starting administration of DARZALEX FASPRO®. Monitor for local reactions and consider symptomatic management
Safety generally consistent with DARZALEX®
In COLUMBA, the safety of DARZALEX FASPRO® was demonstrated to be generally consistent with DARZALEX®.1
Adverse reactions reported in ≥10% of patients treated with either DARZALEX FASPRO® or DARZALEX®1


ARR=administration-related reaction.
aUpper respiratory tract infection includes acute sinusitis, nasopharyngitis, pharyngitis, respiratory syncytial virus infection, respiratory tract infection, rhinitis, rhinovirus infection, sinusitis, and upper respiratory tract infection.
bPneumonia includes lower respiratory tract infection, lung infection, pneumocystis jirovecii pneumonia, and pneumonia.
cFatigue includes asthenia and fatigue.
dSystemic ARRs includes terms determined by investigators to be related to infusion. In clinical trials of DARZALEX FASPRO® and DARZALEX®, and the Prescribing Information for DARZALEX®, the term "infusion-related reactions" was used instead of "systemic ARRs."
eCough includes cough and productive cough.
fDyspnea includes dyspnea and dyspnea exertional.
gOnly Grade 3 adverse reactions occurred.
hGrade 5 adverse reactions occurred.
Serious adverse reactions occurred in 26% of patients who received DARZALEX FASPRO® vs 29% of patients who received DARZALEX®. Fatal reactions occurred in 5% of patients receiving DARZALEX FASPRO®. Fatal adverse reactions occurring in more than 1 patient were general physical health deterioration, septic shock, and respiratory failure. Fatal adverse reactions occurred in 7% of patients receiving DARZALEX®.1,2
In the clinical trial, select laboratory abnormalities worsening from baseline were also found to be consistent in patients receiving either DARZALEX FASPRO® or DARZALEX®1


aDenominator is based on the safety population treated with DARZALEX FASPRO® (n=260) or with DARZALEX® (n=258).
Clinical trials studying efficacy of DARZALEX FASPRO® when used in combination regimens
References:
- DARZALEX FASPRO® [Prescribing Information] Horsham, PA: Janssen Biotech, Inc.
- Mateos M-V, Nahi H, Legiec W, et al. Subcutaneous versus intravenous daratumumab in patients with relapsed or refractory multiple myeloma (COLUMBA): a multicentre, open-label, non-inferiority, randomised, phase 3 trial. Lancet Haematol. 2020;7(5):e370-e380.