36-Month Time to Deepest Response
(Post Hoc Analysis)



You are now viewing a post hoc subset analysis by treatment duration of the MAIA trial.
This information is not included in the current Prescribing Information and has not been evaluated by the FDA. No conclusions should be drawn. These data should be understood in the context of the methodology.
Methodology
In a post hoc subset analysis of patients treated through 36 months*
Treatment duration and long-term outcomes with frontline DARZALEX® + Rd1,2
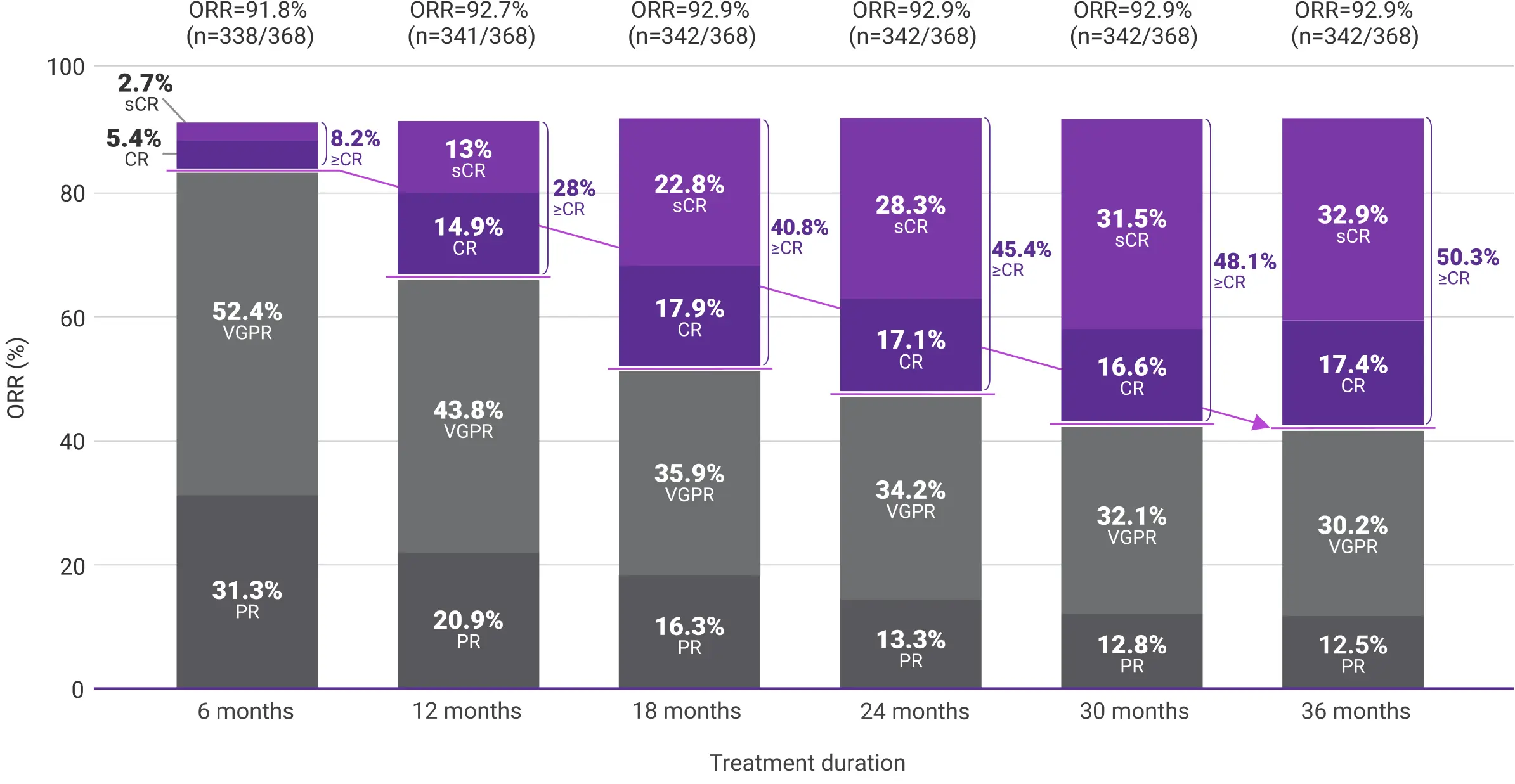
To explore the impact of treatment duration on long-term clinical outcomes, a post hoc analysis evaluated DRd treatment response in 6-month intervals (6, 12, 18, 24, 30, and 36 months).2,3
Response data presented are cumulative deepest response rates achieved by 6, 12, 18, 24, 30, and 36 months, which included 342 out of the 368 ITT population in the DRd arm and 301 out of 369 patients in the Rd arm.2,3
DRd=DARZALEX® (D) + lenalidomide (R) + dexamethasone (d); FDA=U.S. Food and Drug Administration; ITT=intent to treat; Rd=lenalidomide (R) + dexamethasone (d).
*At a median follow-up of 64.5 months.2
In a post hoc subset analysis of patients treated for at least 36 months:
Treatment duration and long-term outcomes with frontline DRd vs Rd1,2
Overall response rates for frontline DARZALEX® + Rd by 36 months2

See an analysis of overall survival based on treatment duration
Explore resultsCR=complete response; DRd=DARZALEX® (D) + lenalidomide (R) + dexamethasone (d); ORR=overall response rate; PR=partial response; Rd=lenalidomide (R) + dexamethasone (d); sCR=stringent complete response; VGPR=very good partial response.
In a post hoc subset analysis of patients treated for at least 36 months:
Treatment duration and long-term outcomes with frontline DRd vs Rd1,2
≥CR by 36 months vs 6 months with Rd1
Overall response rates with frontline Rd by 36 months2
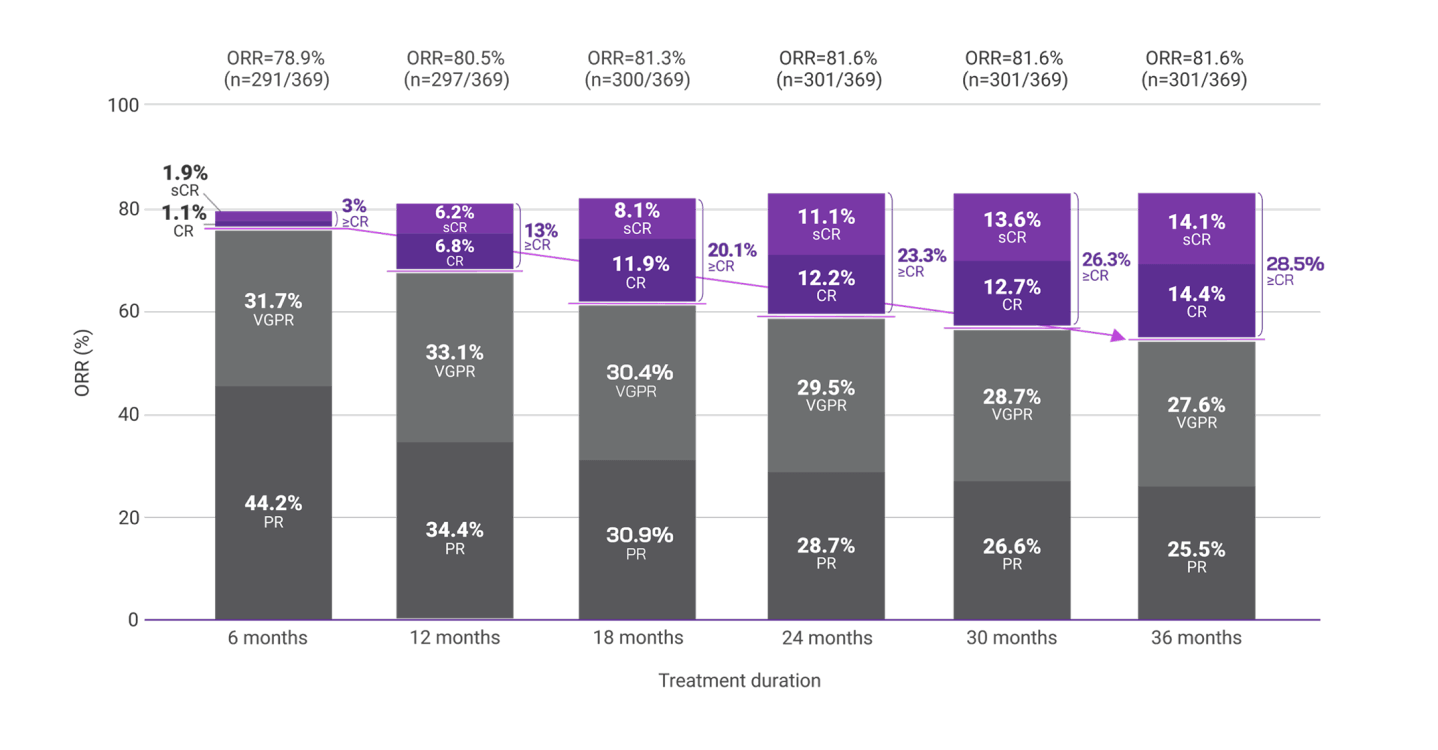
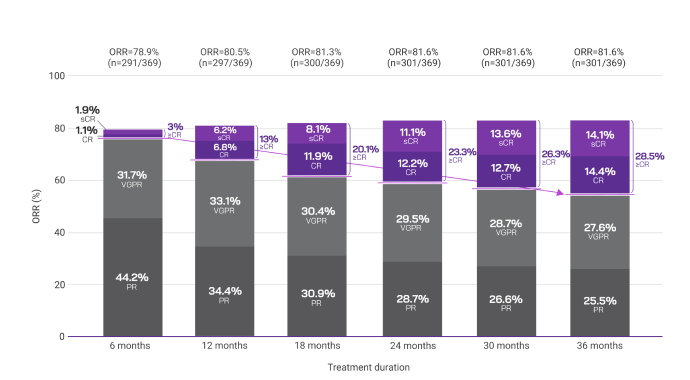
More patients reached a ≥CR with DRd vs Rd (50.3% vs 28.5%, respectively) by 36 months.2
See an analysis of overall survival based on treatment duration
Explore resultsCR=complete response; DRd=DARZALEX® (D) + lenalidomide (R) + dexamethasone (d); ORR=overall response rate; PR=partial response; Rd=lenalidomide (R) + dexamethasone (d); sCR=stringent complete response; VGPR=very good partial response.
References:
Moreau P, Facon T, Usmani SZ, et al. Treatment duration and long-term outcomes with daratumumab in transplant-ineligible newly diagnosed multiple myeloma from the phase 3 MAIA study. Poster presented at: 19th International Myeloma Society (IMS) Annual Meeting; August 25-27, 2022; Los Angeles, CA.
- Data on file. PM-01047. Janssen Biotech, Inc.
Data on file. PM-01145. Janssen Biotech, Inc.

