Safety Over Time


You are now viewing a subset analysis by treatment duration of the MAIA trial. This information is not included in the current Prescribing Information and has not been evaluated by the FDA. No conclusions should be drawn. In the following analysis, treatment-emergent adverse events are presented as observed and should be understood in context with the specific methodology.
Please see MAIA trial design & primary results
Methodology
In patients who continued treatment
An evaluation of frequently reported treatment-emergent adverse events over time from treatment initiation to ~30 months1,2
- Most frequently reported treatment-emergent adverse events (TEAEs) that met the threshold of cumulative any-Grade TEAE ≥30% or Grade 3/4 TEAE ≥10% are presented
- Combined TEAE rates are the sum of the percentages of the most frequently reported TEAEs at each cycle period
- Percentages represent the number of patients with ≥1 TEAE by treatment cycle divided by the total number of patients treated within the treatment window
- Only TEAEs with onset date falling within the cycle intervals were calculated. Each patient was calculated once per preferred term for each cycle interval. The same patient could be calculated in multiple cycle intervals and for multiple preferred terms. TEAEs may not have resolved by the next cycle
- Decrease in adverse event (AE) rates over time from treatment initiation was observed for most AEs in both treatment arms. Cataract tended to increase over time in both treatment arms (see rates on next tab)
- 13% of intent-to-treat (ITT) patients discontinued treatment due to a TEAE with DARZALEX® + Rd (n=364) vs 22% with Rd alone (n=365)1
AE=adverse event; FDA=U.S. Food and Drug Administration; Rd=lenalidomide (R) + dexamethasone (d); TEAE=treatment-emergent adverse event.
In patients who continued treatment
Rates of new-onset AEs decreased over time from treatment initiation to ~30 months with DRd2
Frequently reported adverse events for DRd across Cycles 1-30*
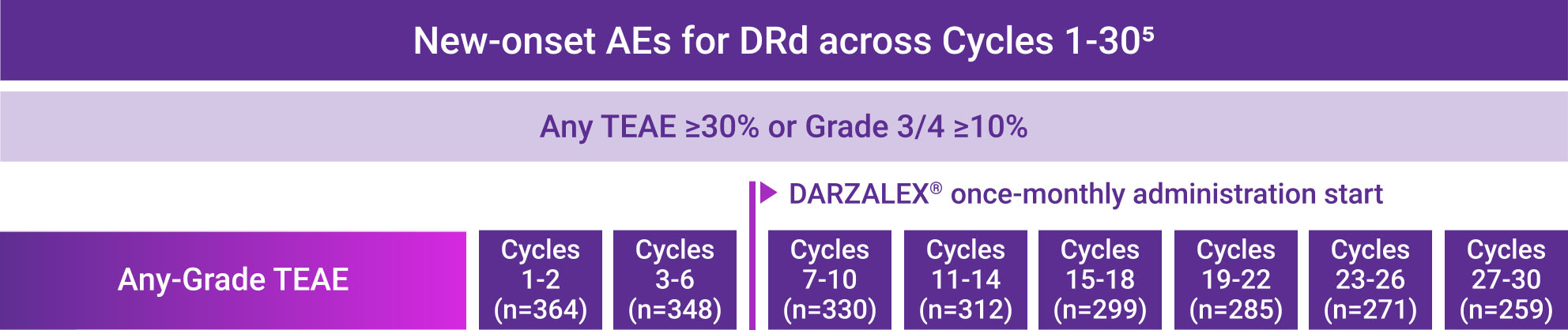
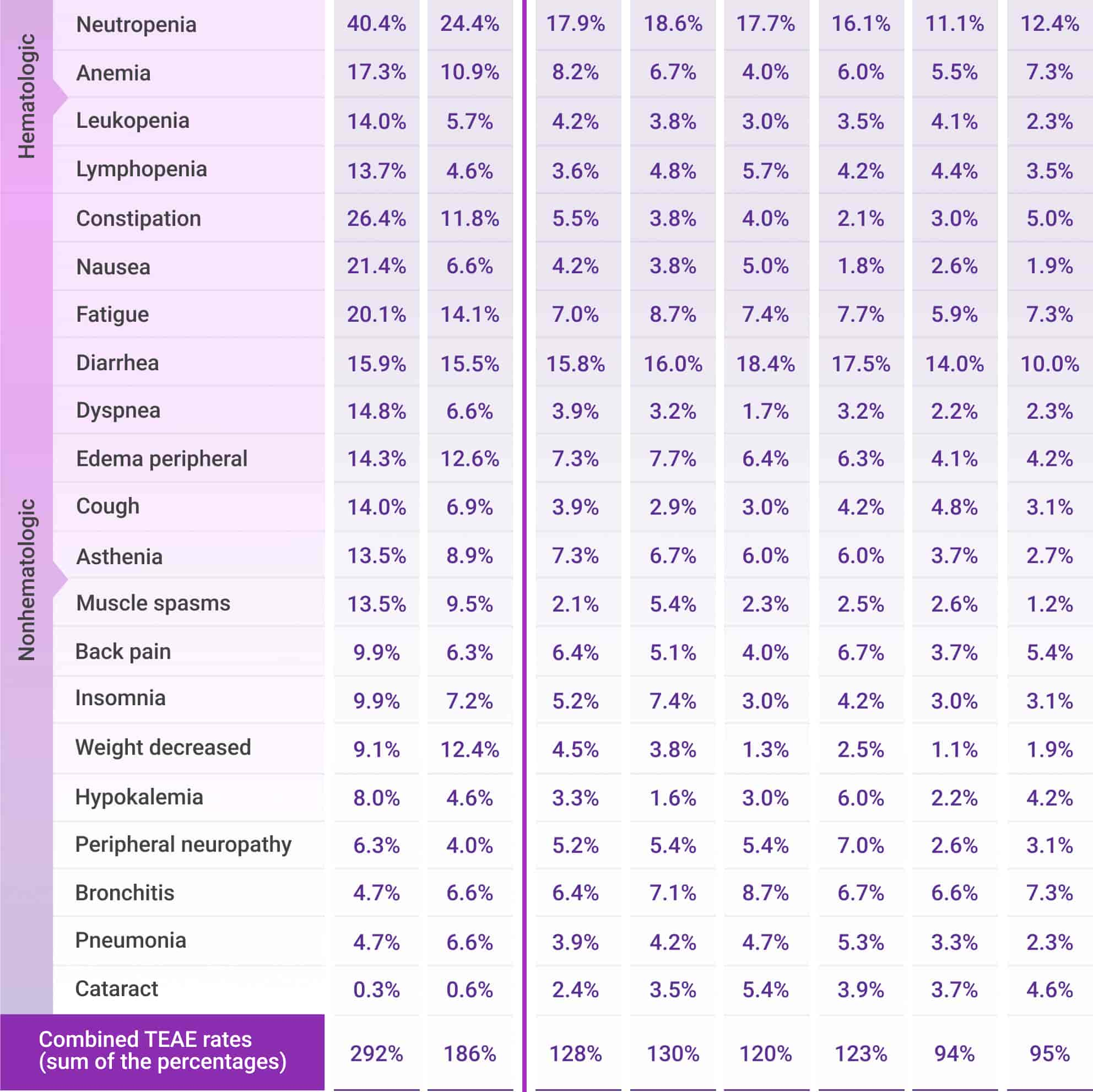


AE=adverse event; DRd=DARZALEX® (D) + lenalidomide (R) + dexamethasone (d); TEAE=treatment-emergent adverse event.
*Cycle=28 days.
In patients who continued treatment
Rates of new-onset AEs decreased over time from treatment initiation to ~30 months with DRd2
Frequently reported adverse events for Rd across Cycles 1-30*

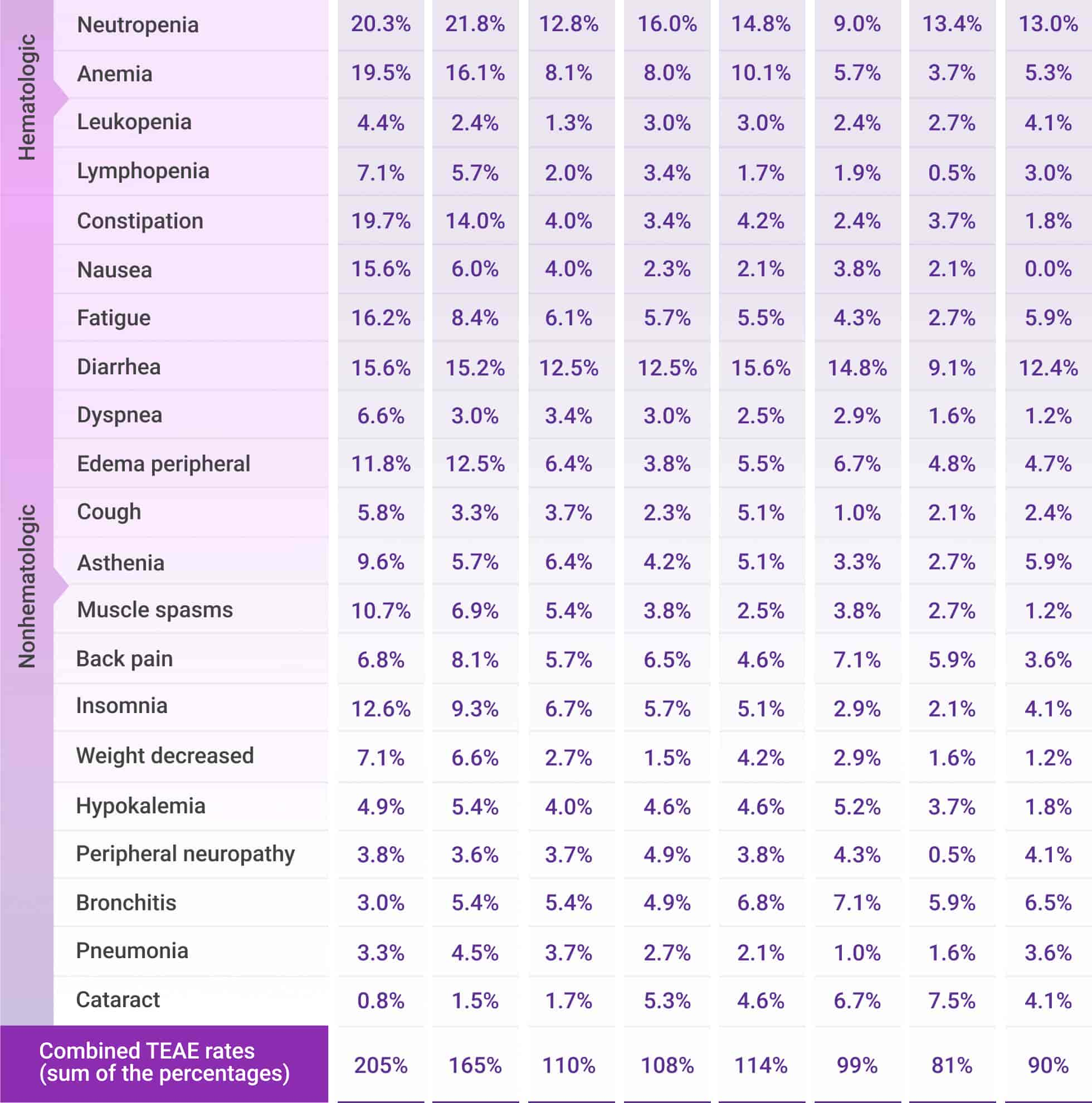


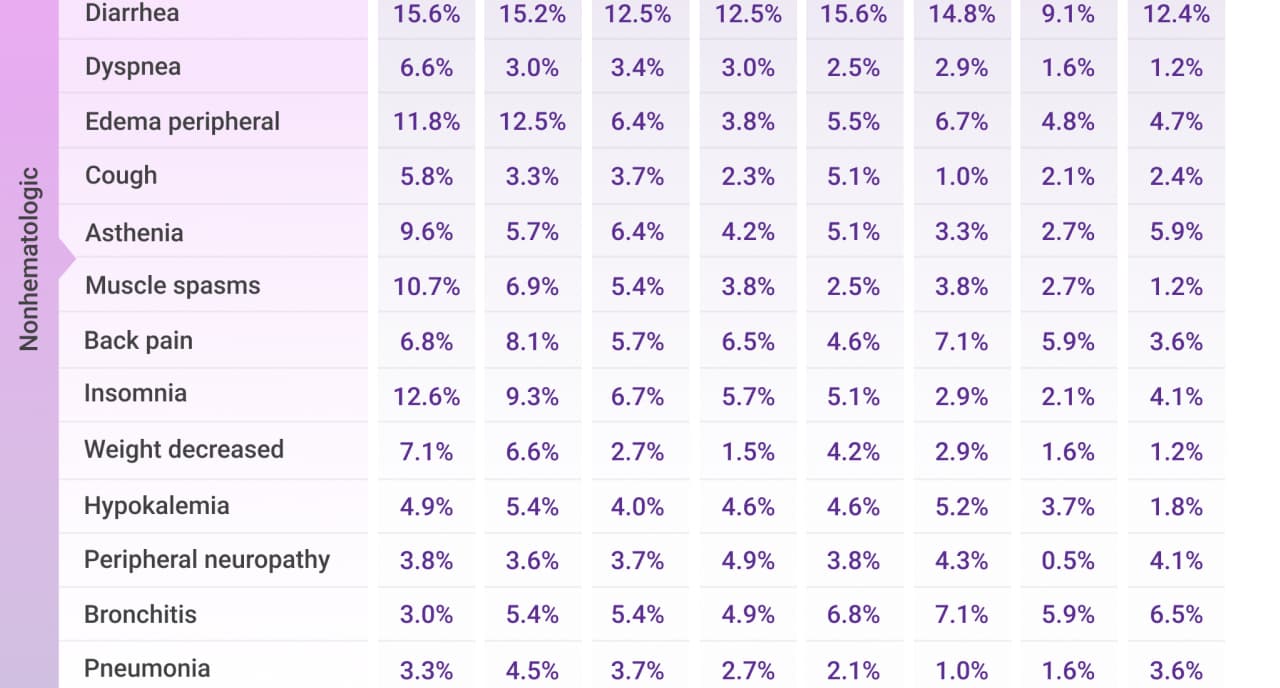
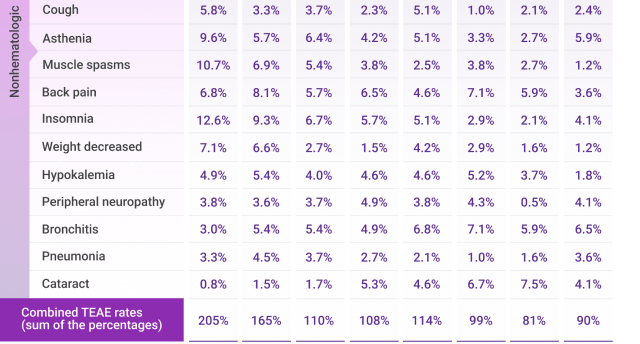
AE=adverse event; Rd=lenalidomide (R) + dexamethasone (d); TEAE=treatment-emergent adverse event.
*Cycle=28 days.
Combined TEAEs
In patients who continued treatment
Combined rates of frequently reported, new-onset AEs decreased over time from treatment initiation to ~30 months with DRd and Rd2
Observed overall AE rates for DRd vs Rd alone (combined rates of most frequent AEs per treatment cycle)2
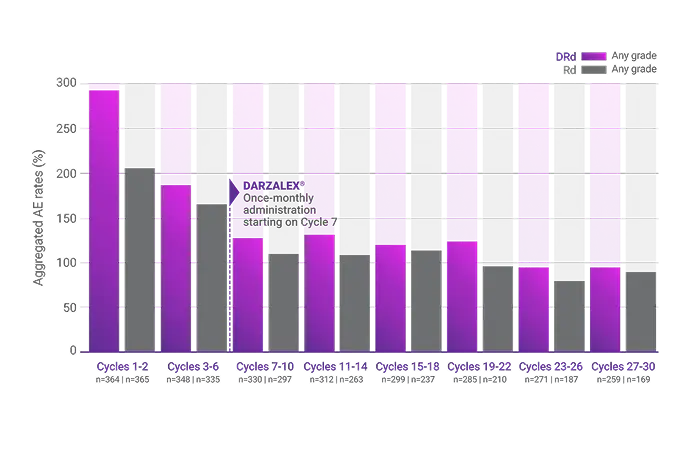
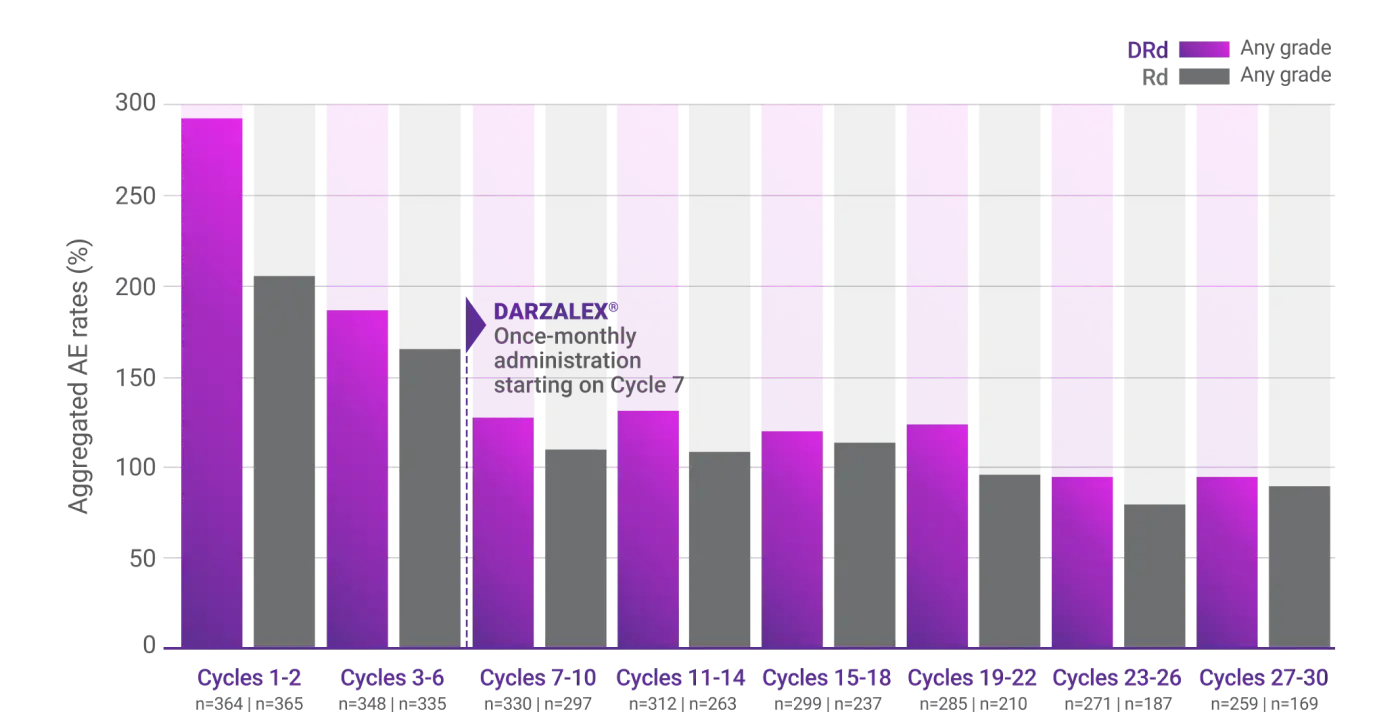
AE=adverse event; DRd=DARZALEX® (D) + lenalidomide (R) + dexamethasone (d); Rd=lenalidomide (R) + dexamethasone (d); TEAE=treatment-emergent adverse event.
References:
Facon T, Kumar SK, Plesner T, et al. Daratumumab, lenalidomide, and dexamethasone versus lenalidomide and dexamethasone alone in newly diagnosed multiple myeloma (MAIA): overall survival results from a randomised, open-label, phase 3 trial. Lancet Oncol. 2021;22(11):1582-1596. doi:10.106/S1470-2045(21)00466-6
Data on file. RF-224245. Janssen Biotech, Inc.

