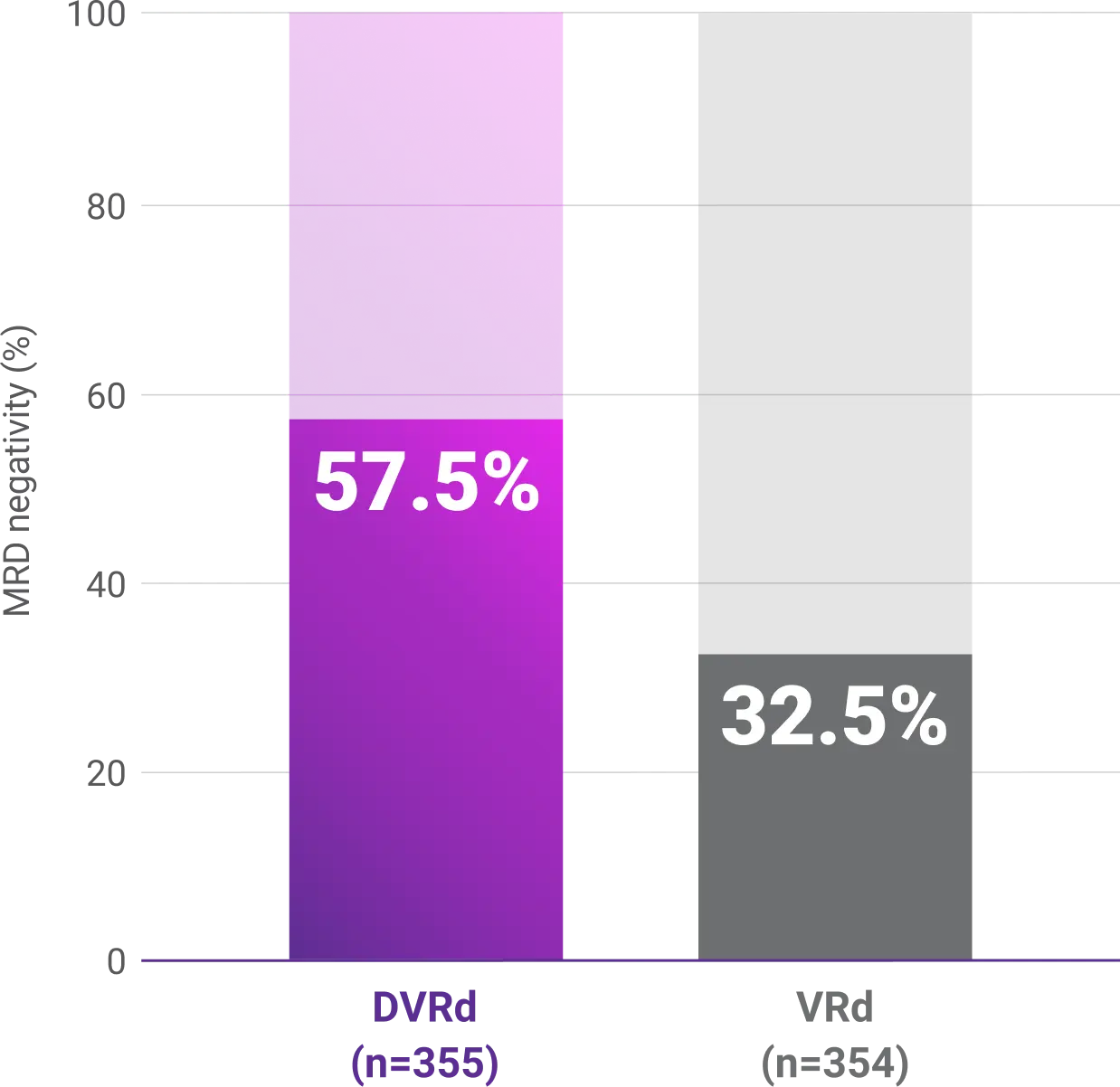
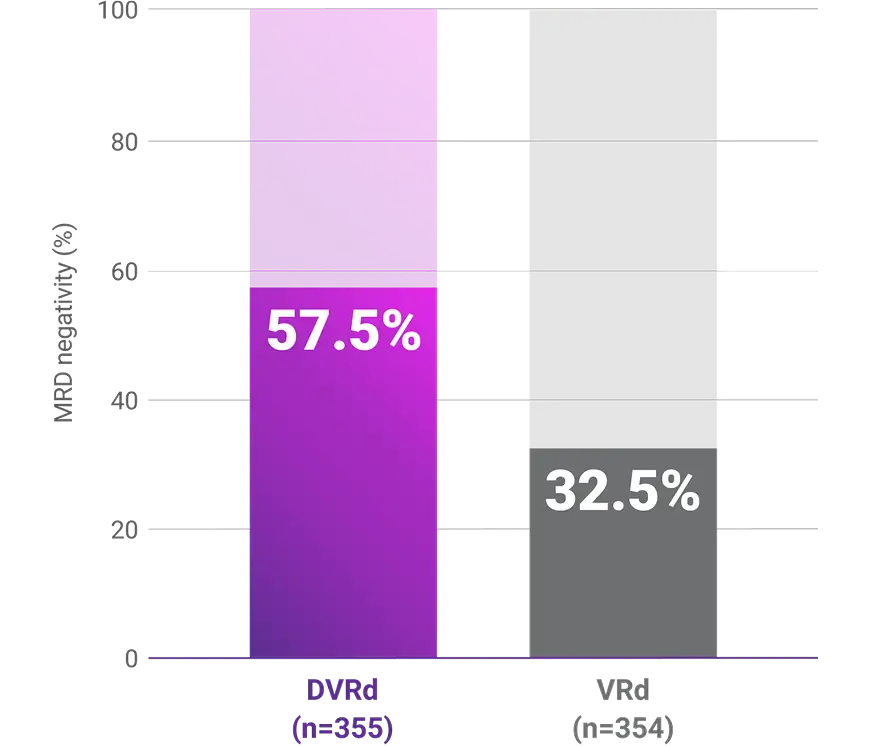
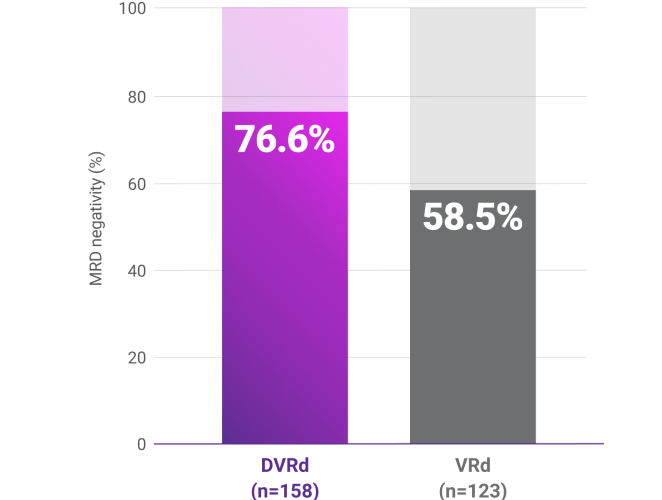
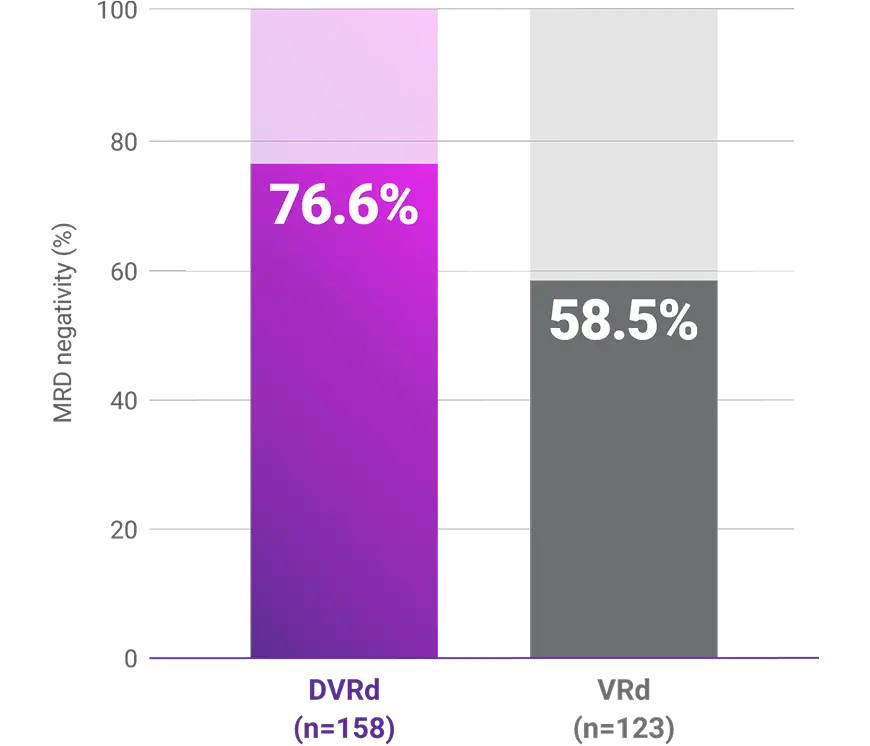
Progression-free survival (PFS)1-3
The median time to reach post-transplant consolidation was 9.9 months in the DVRd arm (range: 0.5-18.5 months).1
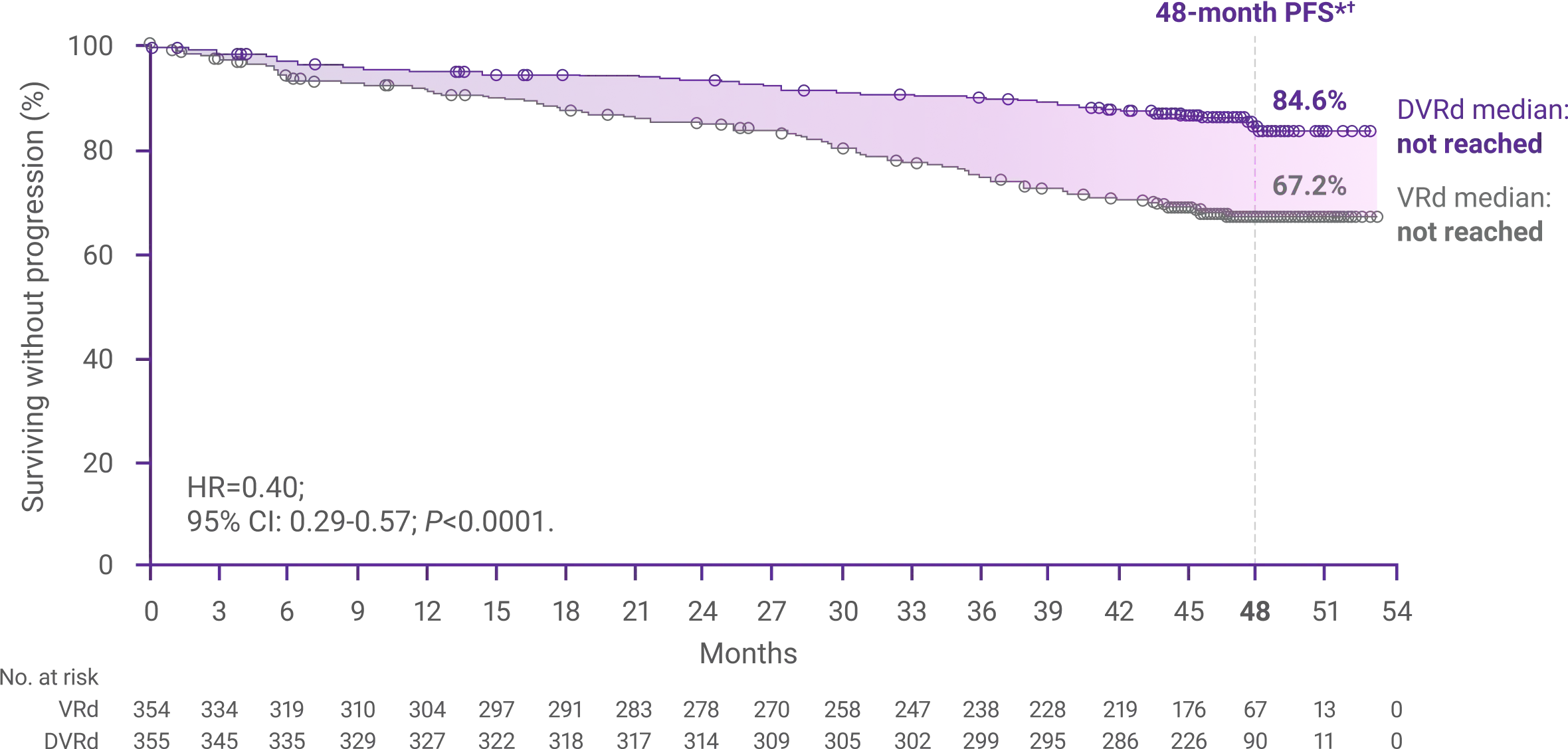
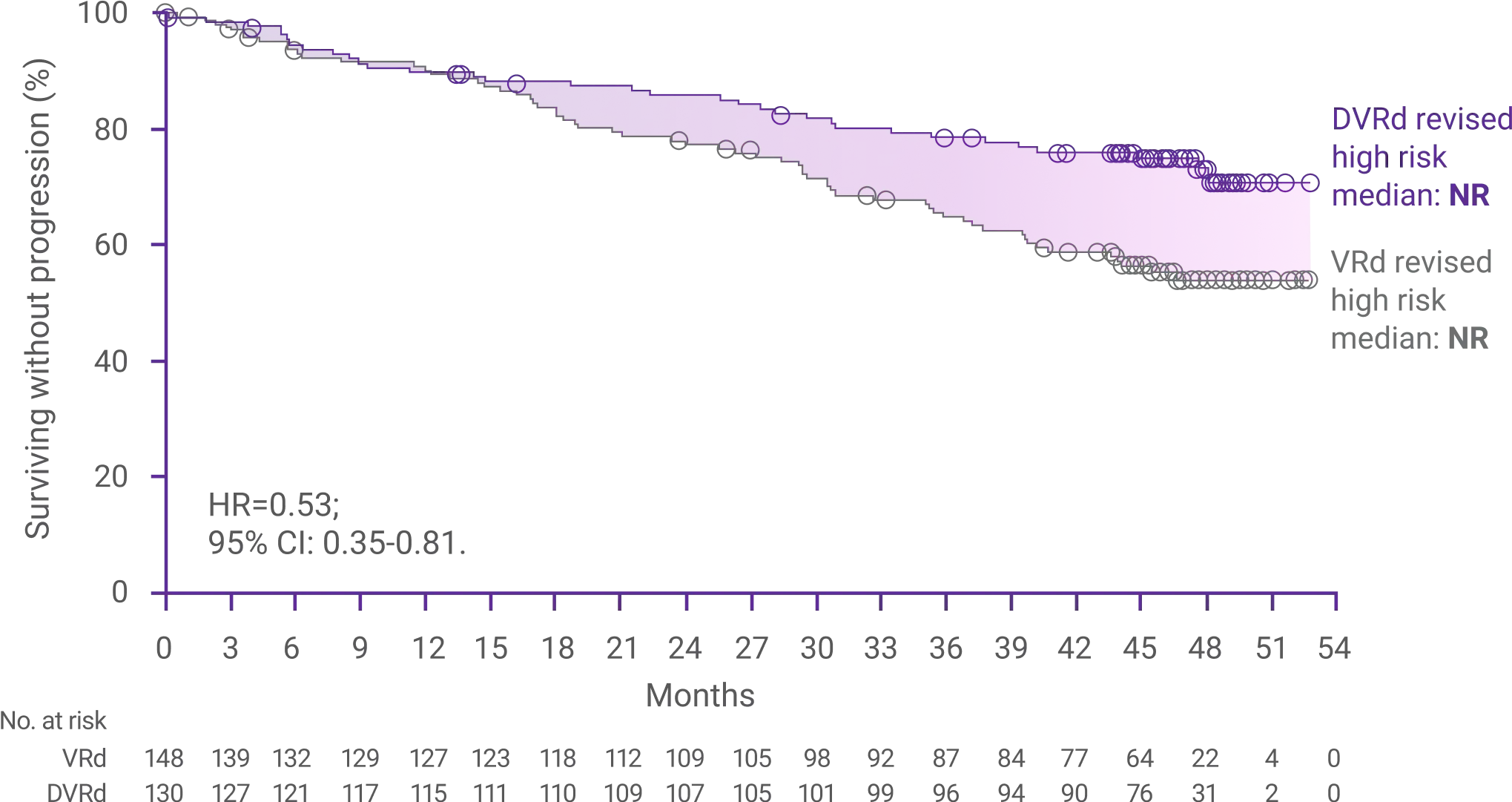
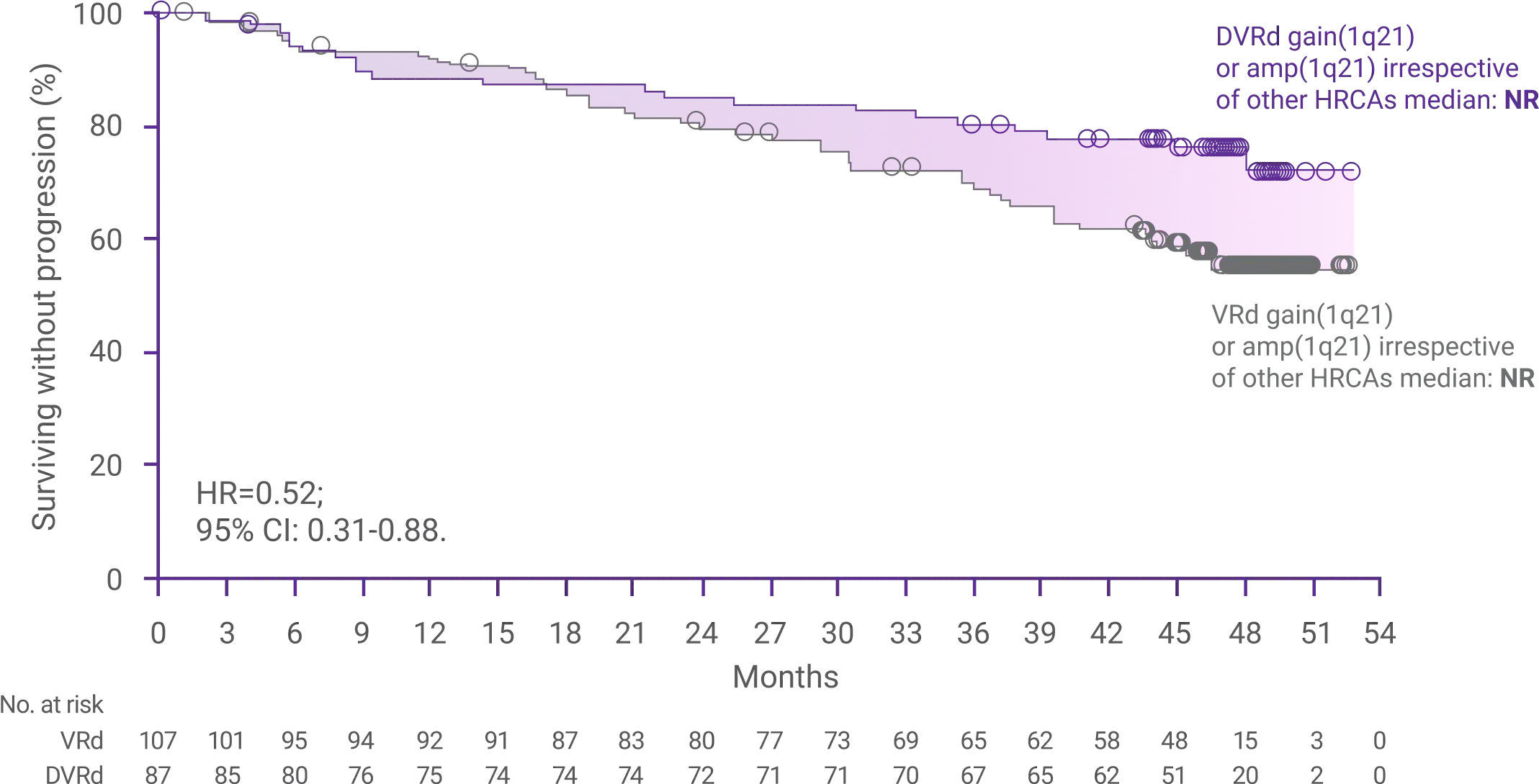
60%reduction in the risk
for disease progression or death with DARZALEX FASPRO®+ VRd vs VRd alone (HR=0.40; 95% CI: 0.29-0.57; P<0.0001)1,2*‡
These results are based on the full treatment regimen and include investigational maintenance of DARZALEX FASPRO® + R following post-transplant consolidation. The trial was not designed to isolate the effect of DARZALEX FASPRO® in the maintenance phase of treatment. The efficacy of DARZALEX FASPRO® + R for maintenance has not been established.1