In the treatment of transplant-eligible patients with newly diagnosed multiple myeloma1:
Demonstrated safety profile through post-transplant consolidation for frontline DARZALEX FASPRO® + VRd1
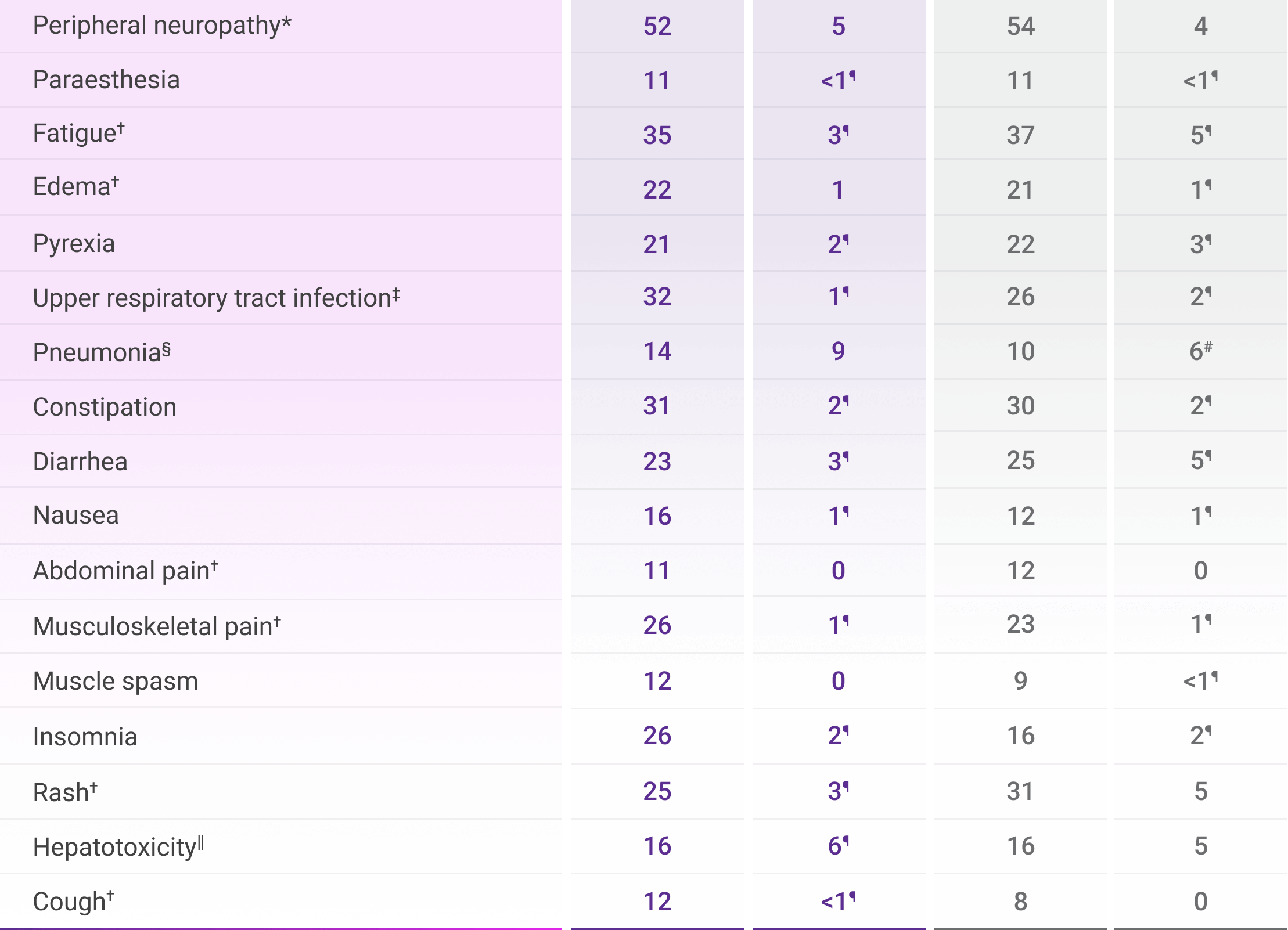
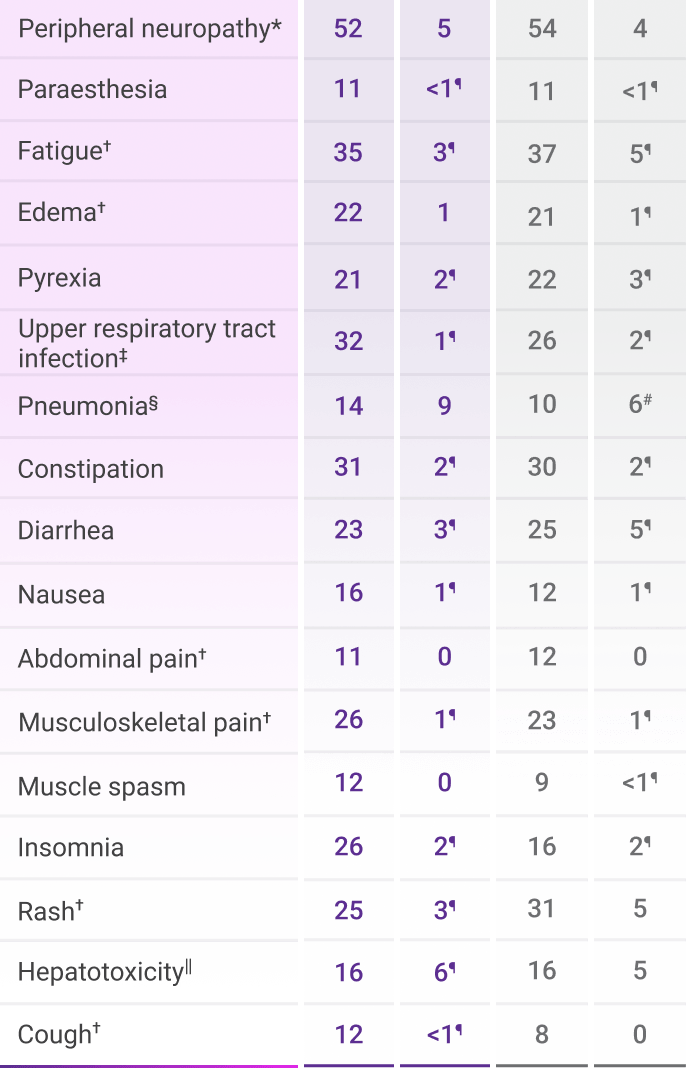
The most common adverse reactions (≥20%) were peripheral neuropathy, fatigue, edema, pyrexia, upper respiratory infection, constipation, diarrhea, musculoskeletal pain, insomnia, and rash.1
In patients who received DVRd1:
- 37% experienced serious adverse reactions
- >5% experienced the most frequent serious adverse reaction: pneumonia (6%)
- 1.7% experienced fatal adverse reactions#
- 2% experienced permanent treatment discontinuation due to an adverse reaction
- An adverse reaction which resulted in permanent discontinuation of DVRd in more than 1 patient included sepsis
ALT=alanine aminotransferase; DVRd=DARZALEX FASPRO® (D) + bortezomib (V) + lenalidomide (R) + dexamethasone (d); VRd=bortezomib (V) + lenalidomide (R) + dexamethasone (d).
*Peripheral neuropathy includes neuropathy peripheral, peripheral motor neuropathy, peripheral sensorimotor neuropathy, and peripheral sensory neuropathy.1
†Includes other related terms.1
‡Upper respiratory tract infection includes fungal pharyngitis, h1n1 influenza, influenza, influenza like illness, laryngitis, nasopharyngitis, oral candidiasis, oropharyngeal candidiasis, parainfluenzae virus infection, pharyngitis, respiratory moniliasis, respiratory syncytial virus infection, respiratory tract infection, respiratory tract infection viral, rhinitis, rhinovirus infection, sinusitis, tonsillitis, upper respiratory tract infection, viral tonsillitis, and viral upper respiratory tract infection.1
§Pneumonia includes bronchopulmonary aspergillosis, lower respiratory tract infection, pneumocystis jirovecii pneumonia, pneumonia, pneumonia bacterial, pneumonia cytomegaloviral, pneumonia influenzal, pneumonia klebsiella, pneumonia legionella, and pneumonia streptococcal.1
||Hepatotoxicity includes alanine aminotransferase increased, aspartate aminotransferase increased, hepatic cytolysis, hepatic failure, hepatic function abnormal, hepatotoxicity, hyperbilirubinemia, hypertransaminasemia, and liver disorder.1
¶Only Grade 3 adverse reactions occurred.1
#Fatal adverse reactions included pneumonia: n=1 (0.3%) in the VRd arm.1
**Based on number of patients with a baseline and post-baseline laboratory value for each laboratory test.1
Adverse reactions were reported in ≥10% of patients who received DVRd through post-transplant consolidation1


Select laboratory abnormalities (≥30%) that worsened from baseline in patients who received DVRd through post-transplant consolidation1

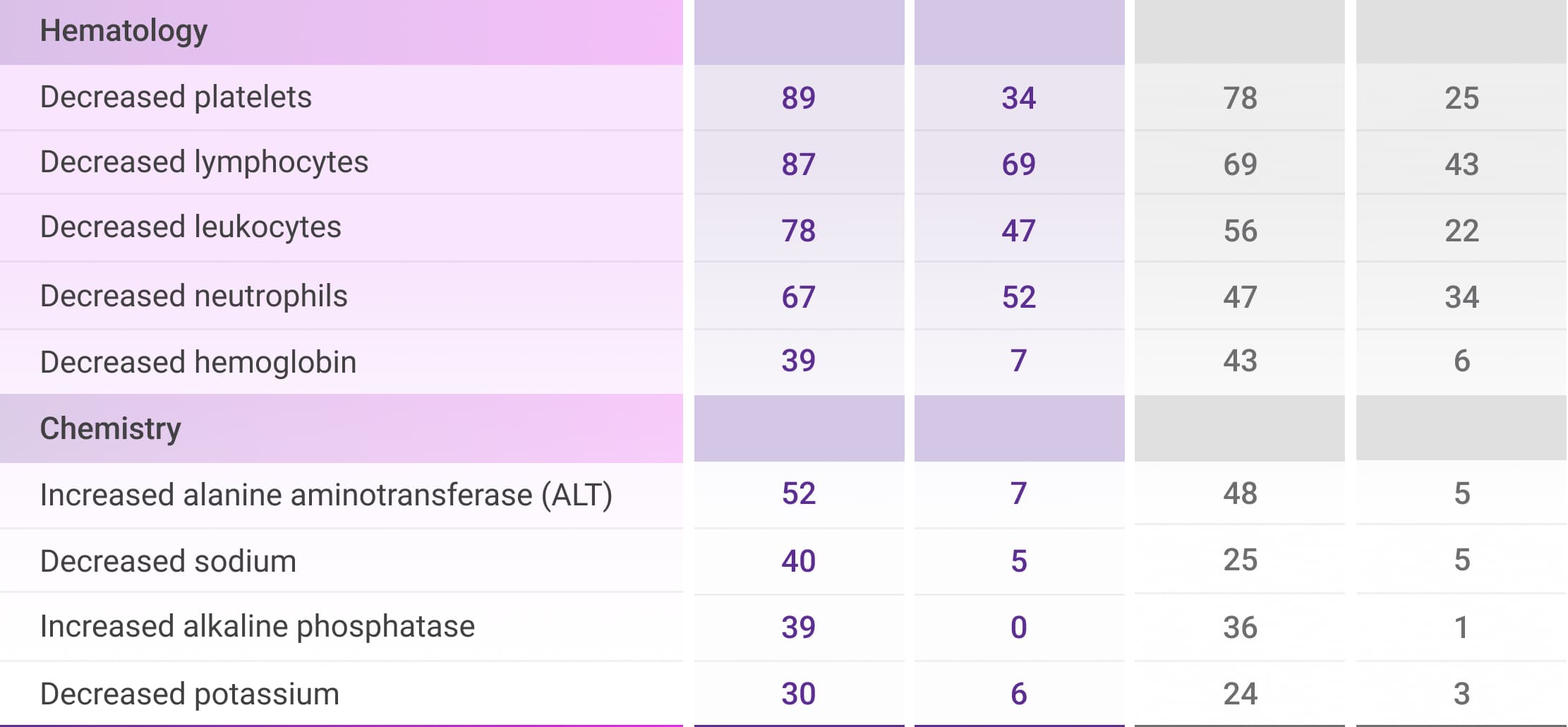
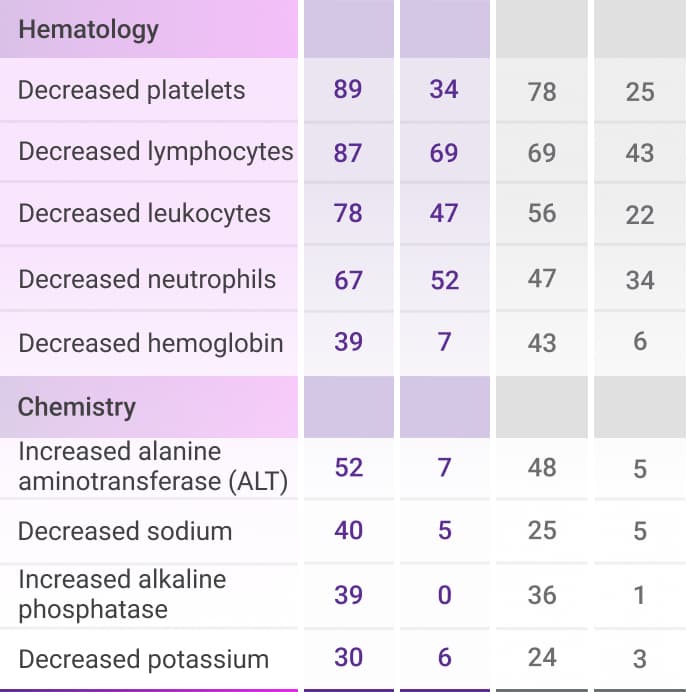
ALT=alanine aminotransferase; DVRd=DARZALEX FASPRO® (D) + bortezomib (V) + lenalidomide (R) + dexamethasone (d); VRd=bortezomib (V) + lenalidomide (R) + dexamethasone (d).
Peripheral neuropathy includes neuropathy peripheral, peripheral motor neuropathy, peripheral sensorimotor neuropathy, and peripheral sensory neuropathy.1
†Includes other related terms.1
‡Upper respiratory tract infection includes fungal pharyngitis, h1n1 influenza, influenza, influenza like illness, laryngitis, nasopharyngitis, oral candidiasis, oropharyngeal candidiasis, parainfluenzae virus infection, pharyngitis, respiratory moniliasis, respiratory syncytial virus infection, respiratory tract infection, respiratory tract infection viral, rhinitis, rhinovirus infection, sinusitis, tonsillitis, upper respiratory tract infection, viral tonsillitis, and viral upper respiratory tract infection.1
§Pneumonia includes bronchopulmonary aspergillosis, lower respiratory tract infection, pneumocystis jirovecii pneumonia, pneumonia, pneumonia bacterial, pneumonia cytomegaloviral, pneumonia influenzal, pneumonia klebsiella, pneumonia legionella, and pneumonia streptococcal.1
||Hepatotoxicity includes alanine aminotransferase increased, aspartate aminotransferase increased, hepatic cytolysis, hepatic failure, hepatic function abnormal, hepatotoxicity, hyperbilirubinemia, hypertransaminasemia, and liver disorder.1
¶Only Grade 3 adverse reactions occurred.1
#Fatal adverse reactions included pneumonia: n=1 (0.3%) in the VRd arm.1
**Based on number of patients with a baseline and post-baseline laboratory value for each laboratory test.1



