PERSEUS Study Design


Approval for the treatment of transplant-eligible patients with newly diagnosed multiple myeloma was based on the results from a phase 3 randomized, multicenter, open-label trial.1,2
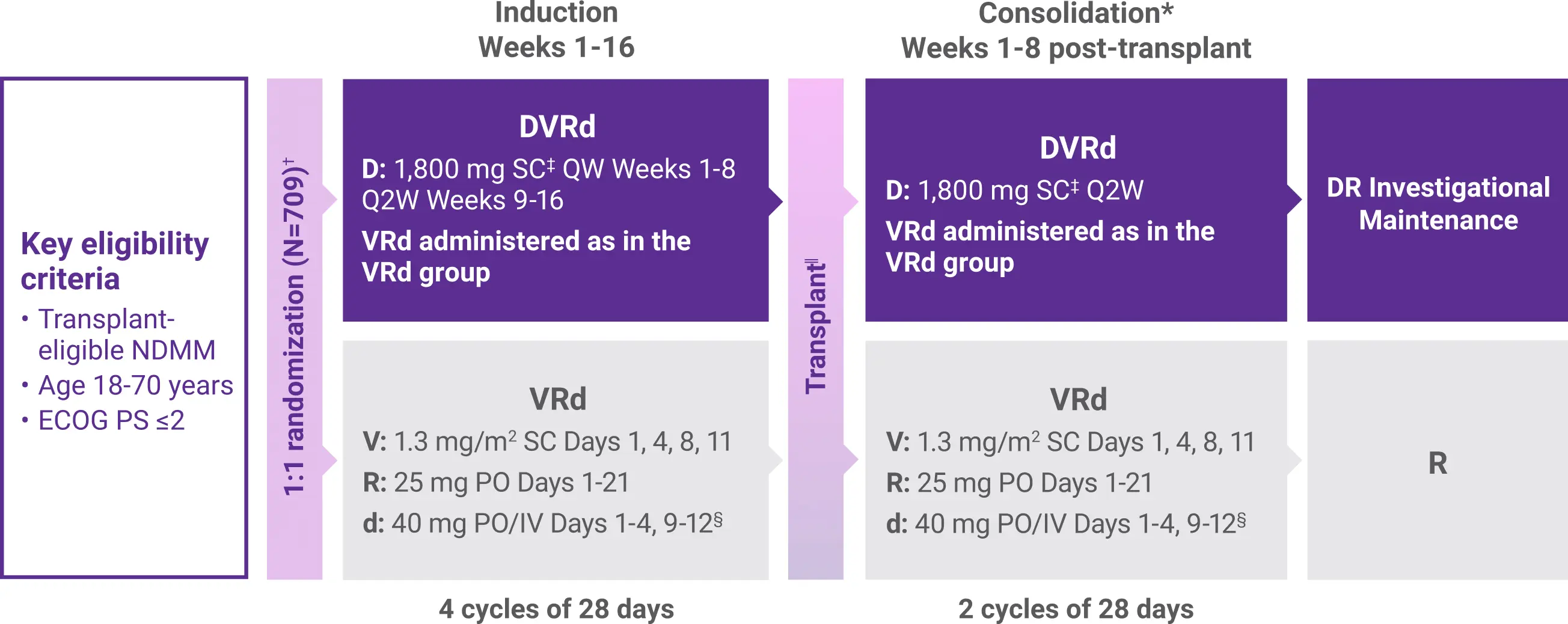
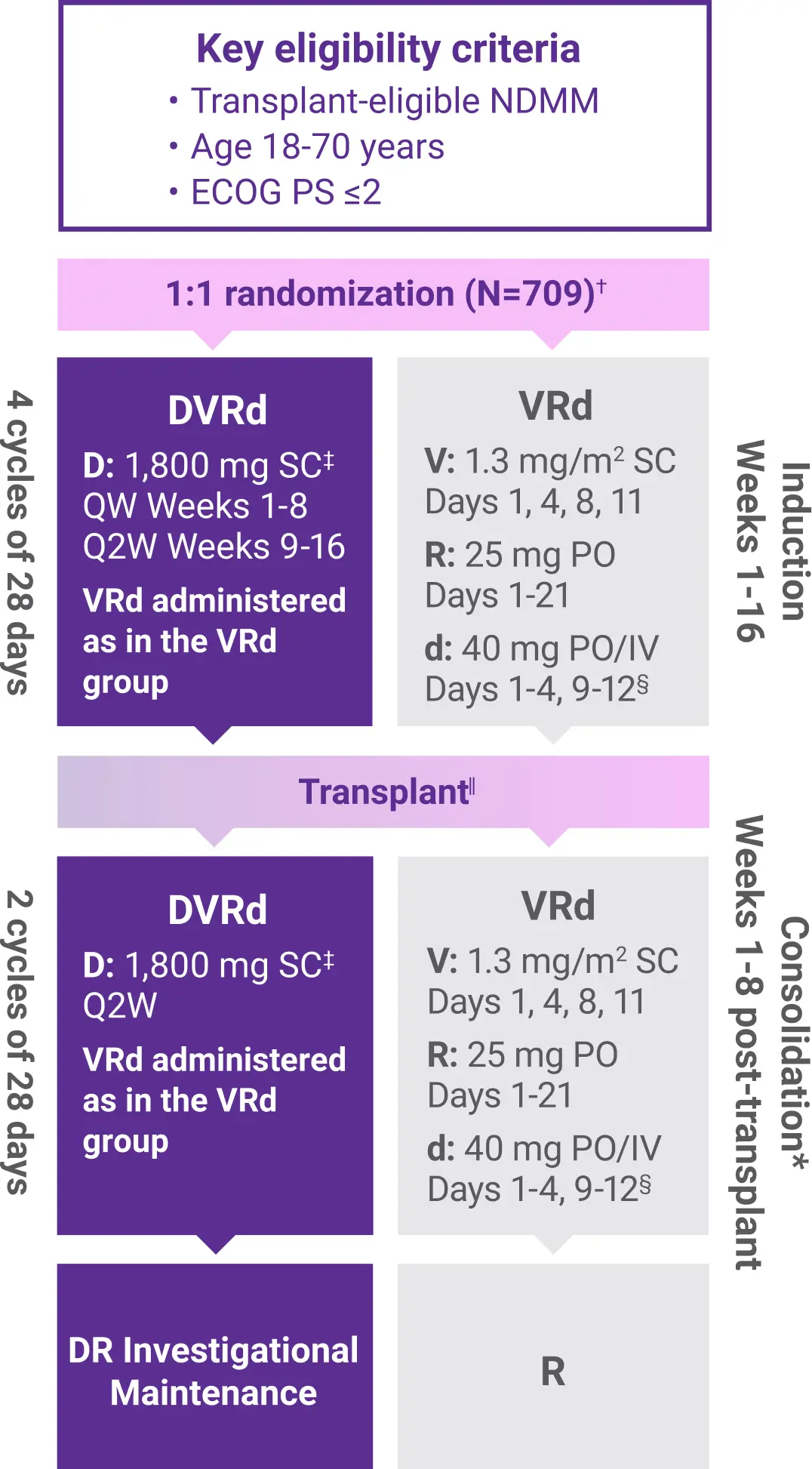
The trial was not designed to isolate the effect of DARZALEX FASPRO® in the maintenance phase of treatment. The efficacy of DARZALEX FASPRO® + R for maintenance has not been established.1
Progression-free survival (PFS) based on International Myeloma Working Group (IMWG) criteria1
Overall response rate (ORR) and minimal residual disease (MRD) negativity rate (next-generation sequencing [NGS]; 10-5)1,2¶
ASCT=autologous stem cell transplant; CR=complete response; d=dexamethasone; D=DARZALEX FASPRO®; DR=DARZALEX FASPRO® (D) + lenalidomide (R); DVRd=DARZALEX FASPRO® (D) + bortezomib (V) + lenalidomide (R) + dexamethasone (d); ECOG PS=Eastern Cooperative Oncology Group performance status; ISS=International Staging System; IV=intravenous; MRD=minimal residual disease; NDMM=newly diagnosed multiple myeloma; PO=by mouth; QW=weekly; Q2W=every 2 weeks; R=lenalidomide; SC=subcutaneous; V=bortezomib; VRd=bortezomib (V) + lenalidomide (R) + dexamethasone (d).
*Within 12 weeks of ASCT, and when engraftment was complete.1
†Stratified by ISS stage and cytogenetic risk.2
‡DARZALEX FASPRO® 1,800 mg co-formulated with rHuPH20 (2,000 U/mL).1
§On the days of DARZALEX FASPRO® injection, the dexamethasone dose was administered orally or intravenously as a pre-injection medication.1
||After Week 16, patients underwent stem cell mobilization, high-dose chemotherapy, and ASCT.1
¶MRD negativity rate was defined as the proportion of patients who achieved both MRD negativity and ≥CR. MRD was assessed using a next-generation sequencing assay (clonoSEQ).1
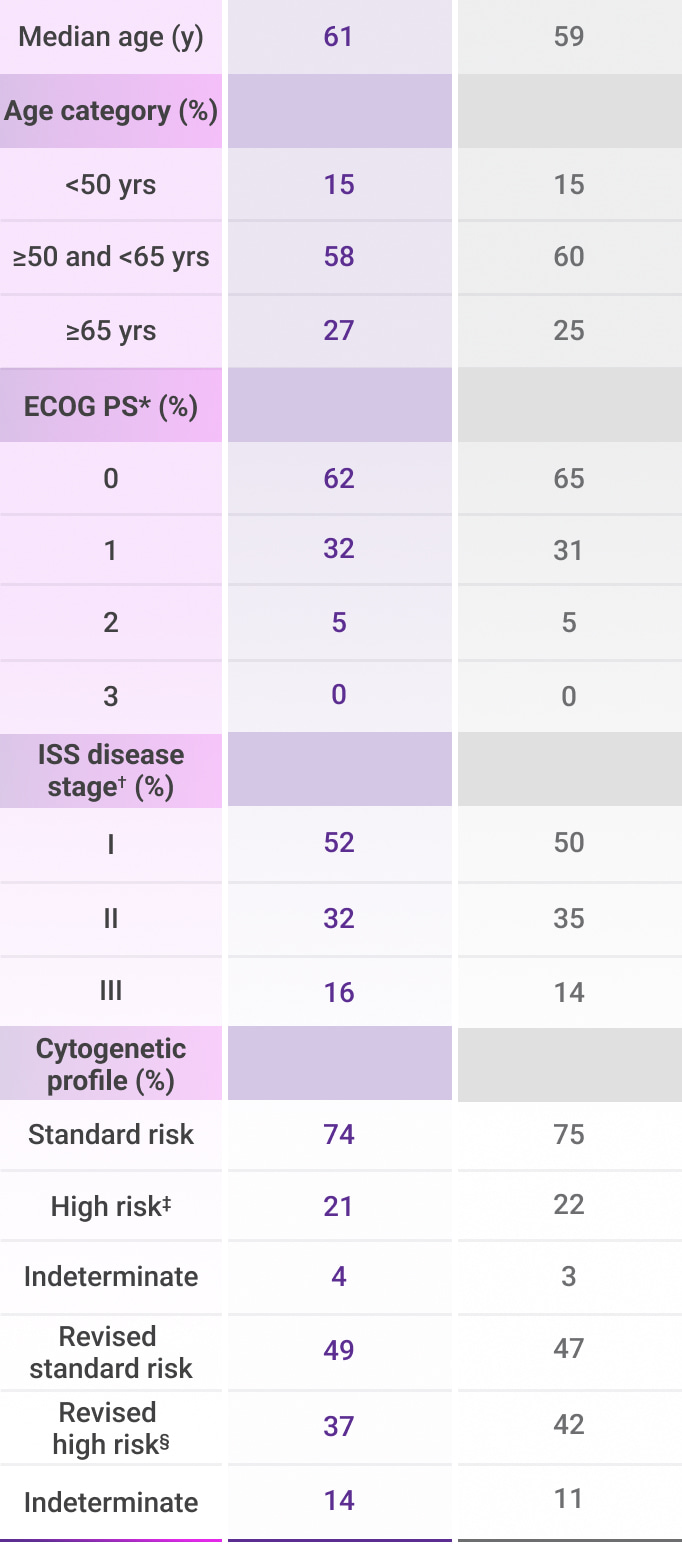
In the treatment of transplant-eligible patients with newly diagnosed multiple myeloma1:
amp=amplification; del=deletion; ECOG PS=Eastern Cooperative Oncology Group performance status; FISH=fluorescence in situ hybridization; ISS=International Staging System; t=translocation; VRd=bortezomib (V) + lenalidomide (R) + dexamethasone (d).
*ECOG PS is scored on a scale from 0 to 5, with 0 indicating no symptoms and higher scores indicating increasing disability.2
†ISS disease stage was based on the combination of serum ß2-microglobulin and albumin levels. Higher stages indicate more advanced disease.2
‡Cytogenetic risk was based on fluorescence in situ hybridization (FISH); high-risk cytogenetics were defined as the presence of one of the following abnormalities: del(17p), t(4;14), or t(14;16).1,2
§Revised cytogenetic high risk was defined as the presence of del(17p), t(4;14), t(14;16), gain(1q21), or amp(1q21).4

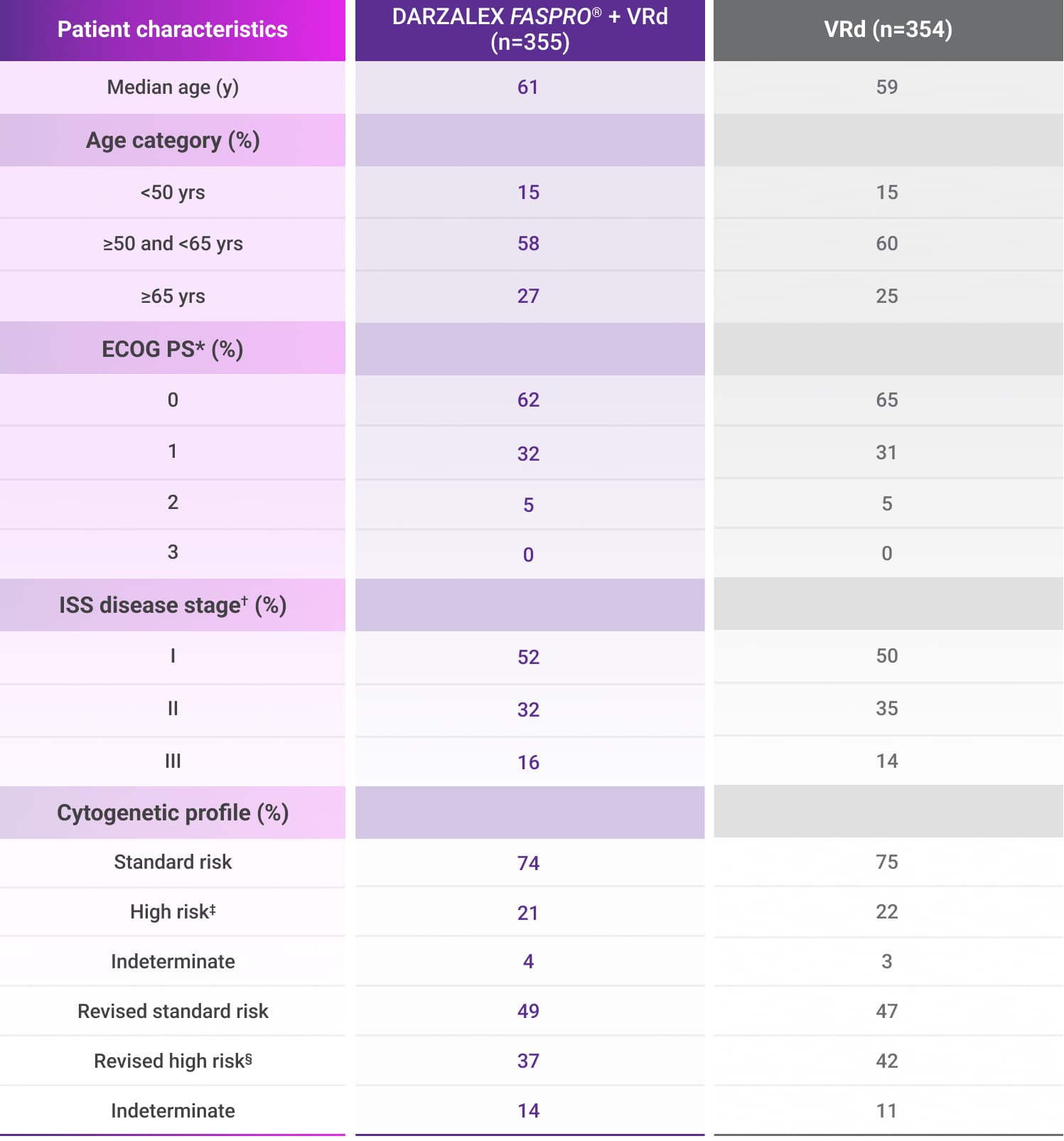
amp=amplification; del=deletion; ECOG PS=Eastern Cooperative Oncology Group performance status; FISH=fluorescence in situ hybridization; ISS=International Staging System; t=translocation; VRd=bortezomib (V) + lenalidomide (R) + dexamethasone (d).
*ECOG PS is scored on a scale from 0 to 5, with 0 indicating no symptoms and higher scores indicating increasing disability.2
†ISS disease stage was based on the combination of serum ß2-microglobulin and albumin levels. Higher stages indicate more advanced disease.2
‡Cytogenetic risk was based on fluorescence in situ hybridization (FISH); high-risk cytogenetics were defined as the presence of one of the following abnormalities: del(17p), t(4;14), or t(14;16).1,2
§Revised cytogenetic high-risk was defined as the presence of del(17p), t(4;14), t(14;16), gain(1q21), or amp(1q21).4
References:
- DARZALEX FASPRO® [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc.
- Sonneveld P, Dimopoulos MA, Boccadoro M, et al. Daratumumab, bortezomib, lenalidomide, and dexamethasone for multiple myeloma. N Engl J Med. 2024;390(4):301-313.
- Sonneveld P, Dimopoulos MA, Boccadoro M, et al. Phase 3 randomized study of daratumumab + bortezomib, lenalidomide, and dexamethasone (VRd) versus VRd alone in patients with newly diagnosed multiple myeloma who are eligible for autologous stem cell transplantation: primary results of the PERSEUS trial. Presented at: 65th American Society of Hematology (ASH) Annual Meeting; December 9-12, 2023; San Diego, CA.
- Dimopoulos MA, Sonneveld P, Rodriguez-Otero P, et al. Daratumumab (DARA)/
bortezomib/lenalidomide/dexamethasone (D-VRd) with D-R maintenance (MAINT) in transplant-eligible (TE) newly diagnosed multiple myeloma (NDMM): analysis of PERSEUS based on cytogenetic risk. Poster presented at: European Hematology Association (EHA) Annual Meeting: June 13-June 16, 2024; Madrid, Spain. - Data on file. PM-01117. Janssen Biotech, Inc.

