Efficacy Results (DRd)


After 28 months of follow-up:
Frontline DARZALEX® + Rd significantly reduced the risk of progression in newly diagnosed, transplant-ineligible patients1*
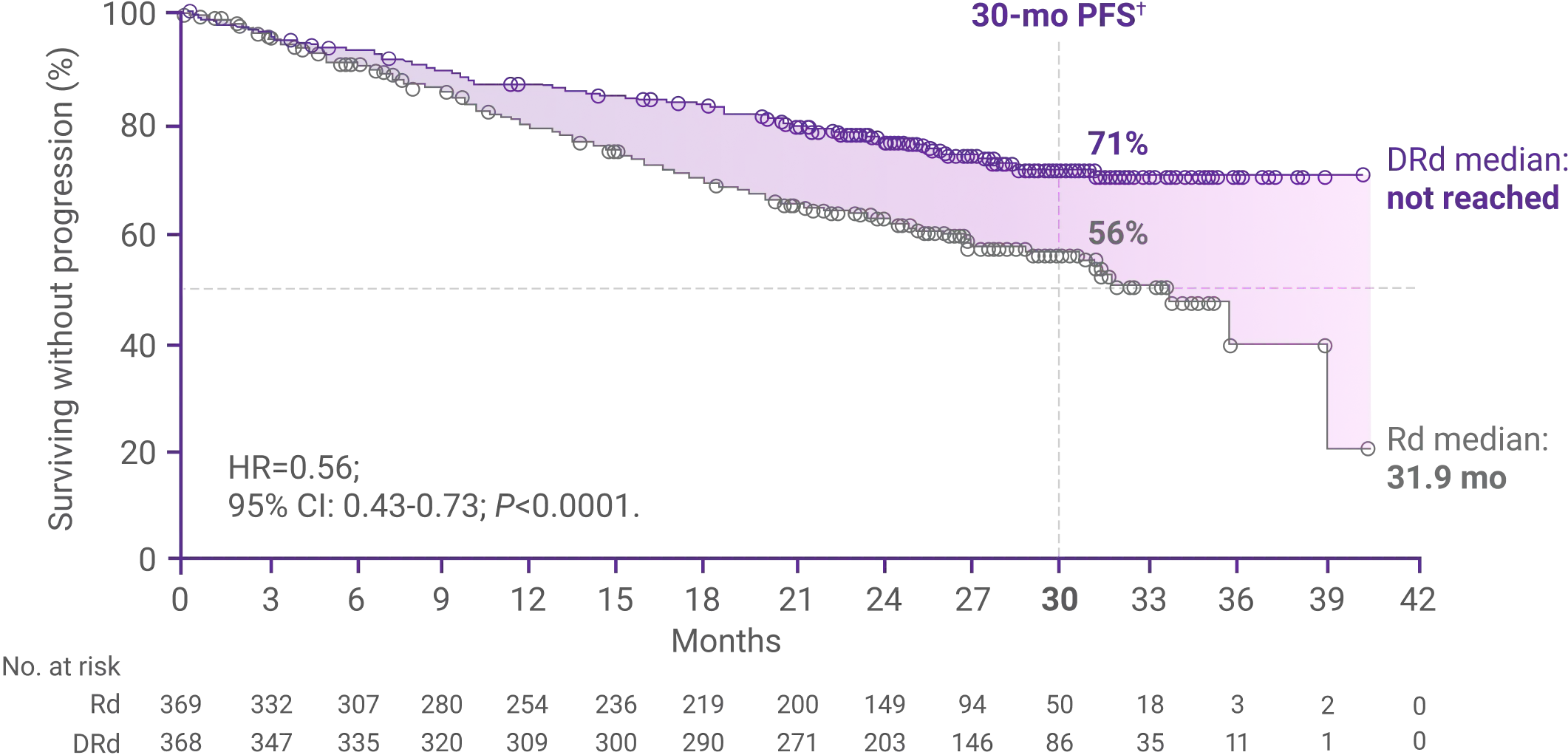
Progression-free survival (PFS) after 28 months1,2
Median PFS was not reached with DRd vs 31.9 months with Rd alone (HR=0.56; 95% CI: 0.43-0.73; P<0.0001)1*†

44%reduction in the risk
of disease progression or death with DRd vs Rd alone
70.6% of patients had not progressed with DRd vs 55.6% with Rd alone at 30 months (DRd: 95% CI: 65.0-75.4; Rd: 95% CI: 49.5-61.3)2†
View 64-month follow-upCI=confidence interval; DRd=DARZALEX® (D) + lenalidomide (R) + dexamethasone (d); HR=hazard ratio; PFS=progression-free survival; Rd=lenalidomide (R) + dexamethasone (d).
*Median follow-up was 28 months (range: 0–41.4 months).1,2
†Kaplan-Meier estimate.2
After 64 months of follow-up:
Patients were progression-free longer with frontline DARZALEX® + Rd vs Rd alone1*
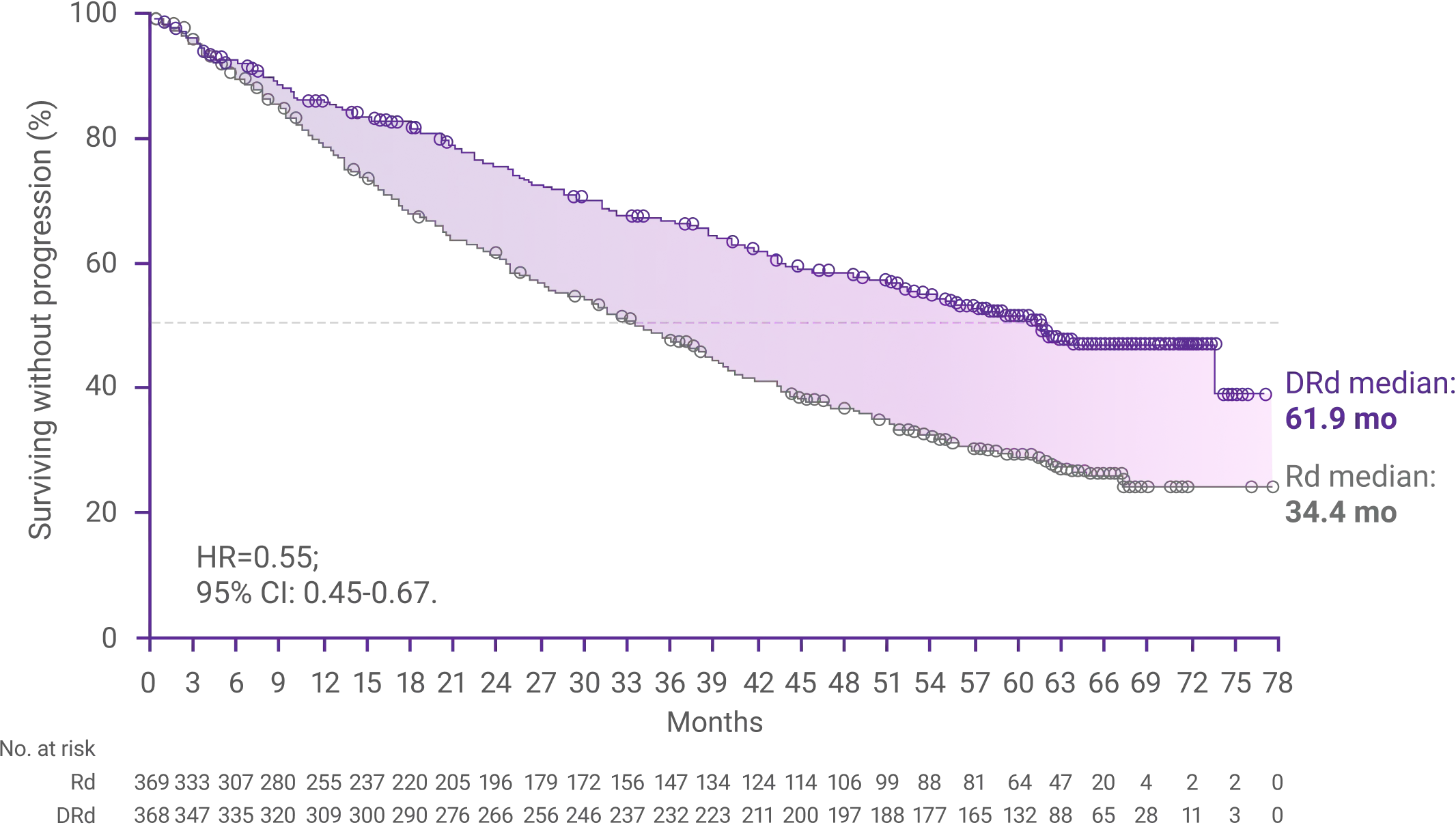
Progression-free survival (PFS)1,3
Median PFS was 61.9 months (95% CI: 54.8-NE) with DRd vs 34.4 months (95% CI: 29.6-39.2) with Rd alone1

45%reduction in the risk
of disease progression or death with DRd vs Rd alone
52.1% of patients in the DRd arm had not progressed at 60 months vs 29.6% of patients in the Rd arm (HR=0.55, 95% CI: 0.45-0.67)3
CI=confidence interval; DRd=DARZALEX® (D) + lenalidomide (R) + dexamethasone (d); HR=hazard ratio; NE=not estimable; PFS=progression-free survival; Rd=lenalidomide (R) + dexamethasone (d).
*Median follow-up was 64 months.1
After 56 months of follow-up:
Frontline DARZALEX® + Rd significantly reduced the risk of death vs Rd alone1*
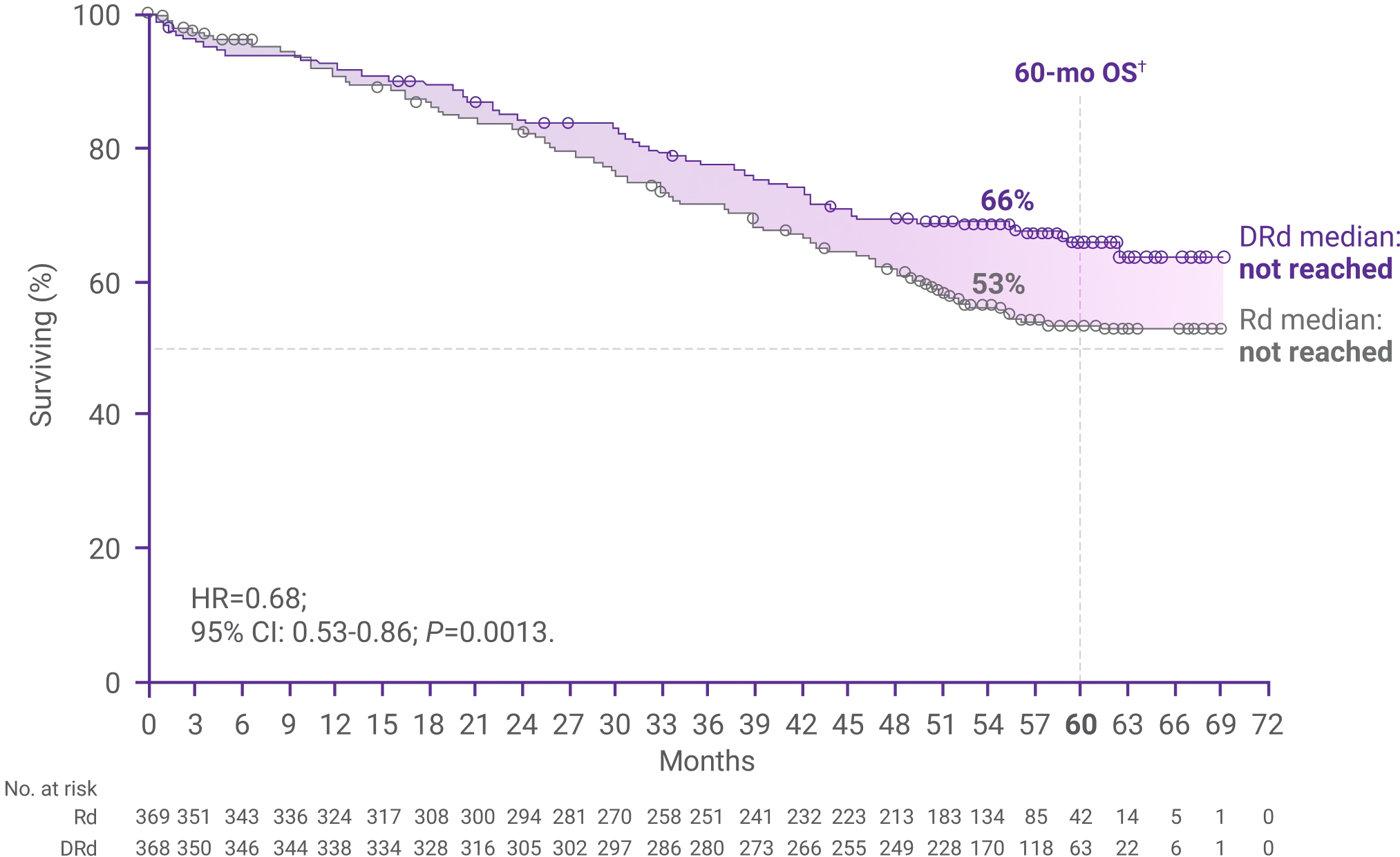
Overall survival after 56 months1,4
Median OS was not reached with DRd and Rd alone (HR=0.68; 95% CI: 0.53-0.86; P=0.0013)1*

32%reduction in the risk
of death with DRd vs Rd alone
66% of patients were still alive with DRd vs 53% with Rd alone at 60 months (DRd: 95% CI: 60.8-71.3; Rd: 95% CI: 47.2-58.6)4*†
View 89-month OSDARZALEX® + Rd is the only anti-CD38 triplet regimen with proven overall survival results1,4
CD38=cluster of differentiation 38; CI=confidence interval; DRd=DARZALEX® (D) + lenalidomide (R) + dexamethasone (d); HR=hazard ratio; IQR=interquartile range; OS=overall survival; Rd=lenalidomide (R) + dexamethasone (d).
*Median follow-up was 56.6 months in the DRd group (IQR: 53.0-60.1 months) and 55.9 months in the Rd group (IQR: 52.5-59.4 months).4
†Kaplan-Meier estimate.4
After 89 months of follow-up:
Patients lived longer with frontline DARZALEX® + Rd vs Rd alone1,5*†
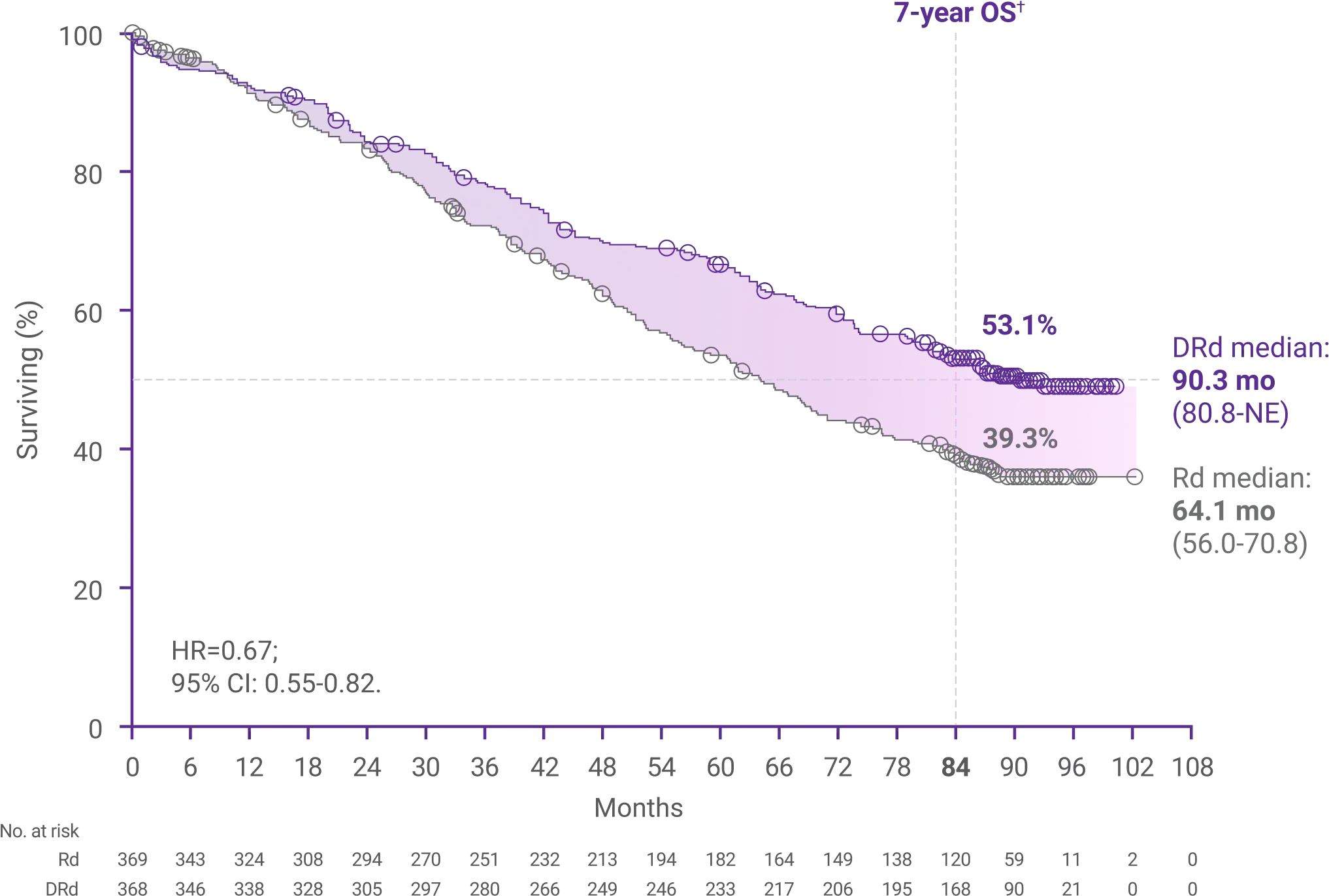
Overall survival after 89 months1,5

33%reduction in the risk
of death with DARZALEX® + Rd vs Rd alone
53.1% of patients in the DRd arm were still alive at 89 months vs 39.3% of patients in the Rd arm (HR=0.67; 95% CI: 0.55-0.82)
CI=confidence interval; DRd=DARZALEX® (D) + lenalidomide (R) + dexamethasone (d); HR=hazard ratio; NE=not estimable; OS=overall survival; Rd=lenalidomide (R) + dexamethasone (d).
*Median follow-up was 89.3 months (range: 0-102.2 months).5
†Kaplan-Meier estimate.5
Deeper responses for a strong start. Durable responses to keep your patients moving forward1,2*
Overall response rates (ORRs)
93% ORR was achieved with DARZALEX® + Rd after 28 months of follow-up1,2*


Depth
DRd nearly doubled the number of patients who achieved CR or better vs Rd alone.1
- sCR more than doubled with DRd (30%) vs Rd alone (13%)
Durability
Median duration of response had not yet been reached with DRd vs 34.7 months (95% CI: 30.8 to not estimable) for Rd alone.1
Speed of Response
Median time to response was 1.05 months for both DRd (range: 0.2–12.1 months) and Rd alone (range: 0.3–15.3 months).1
View 28-month MRD negativity dataCI=confidence interval; CR=complete response; DRd=DARZALEX® (D) + lenalidomide (R) + dexamethasone (d); MRD=minimal residual disease; ORR=overall response rate; PR=partial response; Rd=lenalidomide (R) + dexamethasone (d); sCR=stringent complete response; VGPR=very good partial response.
*Median follow-up was 28 months (range: 0–41.4 months).1,2
‡sCR is CR plus normal free light chain ratio and the absence of clonal cells in bone marrow as assessed by immunohistochemistry or immunofluorescence.2
After 28 months of follow-up:
Patients achieved MRD negativity with frontline DARZALEX® + Rd at a rate 3x greater than Rd alone1,2*†
3.4x greater MRD negativity (24%) with DRd at a median follow-up of 28 months1


Superior MRD rates vs Rd alone
- 24% of patients were minimal residual disease (MRD)-negative with DRd (95% CI: 19.9-28.9)1
- 7% of patients were MRD-negative with Rd (95% CI: 4.9-10.5)1
MRD was based on a sensitivity threshold of 10–5 using a next-generation sequencing assay (clonoSEQ® ).1
View 64-month ORR dataCI=confidence interval; DRd=DARZALEX® (D) + lenalidomide (R) + dexamethasone (d); MRD=minimal residual disease; Rd=lenalidomide (R) + dexamethasone (d).
*Median follow-up was 28 months (range: 0.0-41.4 months).2
†MRD negativity was defined as undetectable levels of multiple myeloma cells by bone marrow aspirate at any time point after the randomization and before disease progression or start of subsequent therapy, and in the trial was assessed by means of next-generation sequencing assay at a sensitivity threshold of 10–5 via bone marrow aspirate, collected at initial trial screening, at the time of confirmation of complete response or stringent complete response, and thereafter at 12, 18, 24, and 30 months.2
Cumulative response rates after 64.5 months of follow-up3*
You are now viewing a follow-up analysis of the MAIA study. This information is not included in the current Prescribing Information and has not been evaluated by the FDA. The follow-up analysis was not adjusted for multiplicity and conclusions should not be drawn.
Cumulative ORRs3
93% ORR was achieved with DARZALEX® + Rd after 64.5 months of follow-up3*


Depth3
1.7x more patients receiving DRd achieved CR or better vs Rd alone
- More than doubled sCR‡: 36% with DRd vs 16% with Rd alone
The follow-up analysis was not adjusted for multiplicity and no conclusions should be drawn.
CR=complete response; DRd=DARZALEX® (D) + lenalidomide (R) + dexamethasone (d); FDA=U.S. Food and Drug Administration; ORR=overall response rate; PR=partial response; Rd=lenalidomide (R) + dexamethasone (d); sCR=stringent complete response; VGPR=very good partial response.
*Median follow-up was 64.5 months.3
‡sCR is CR plus normal free light chain ratio and the absence of clonal cells in bone marrow as assessed by immunohistochemistry or immunofluorescence.2
PFS numerically favored frontline DARZALEX® + Rd in most subgroups vs Rd alone after 28 months of follow-up2*
These analyses are not included in the Prescribing Information for DARZALEX®. These analyses were not adjusted for multiple comparisons.
There are insufficient numbers of patients per subgroup to make definitive conclusions of efficacy among the subgroups.
Progression-free survival

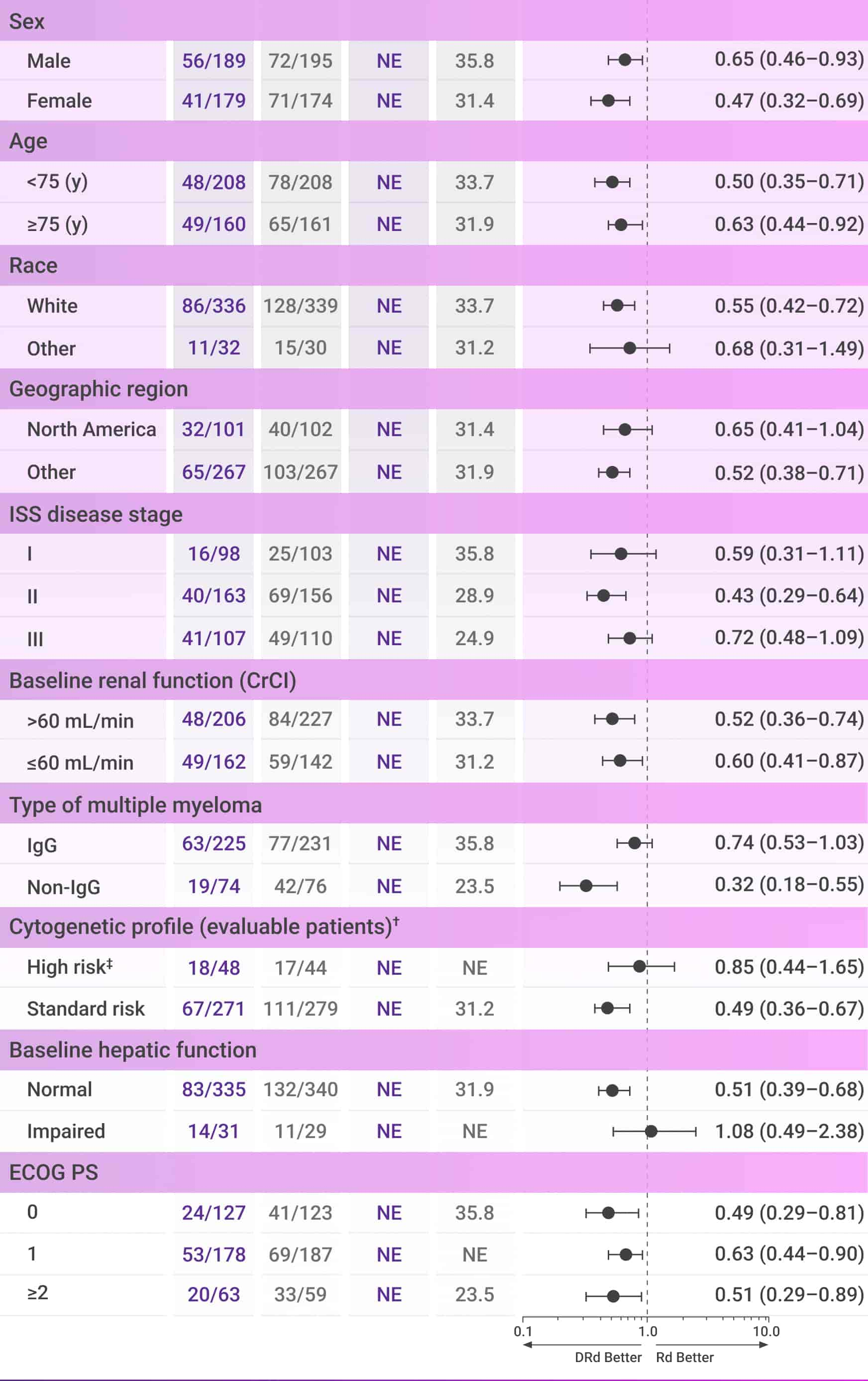

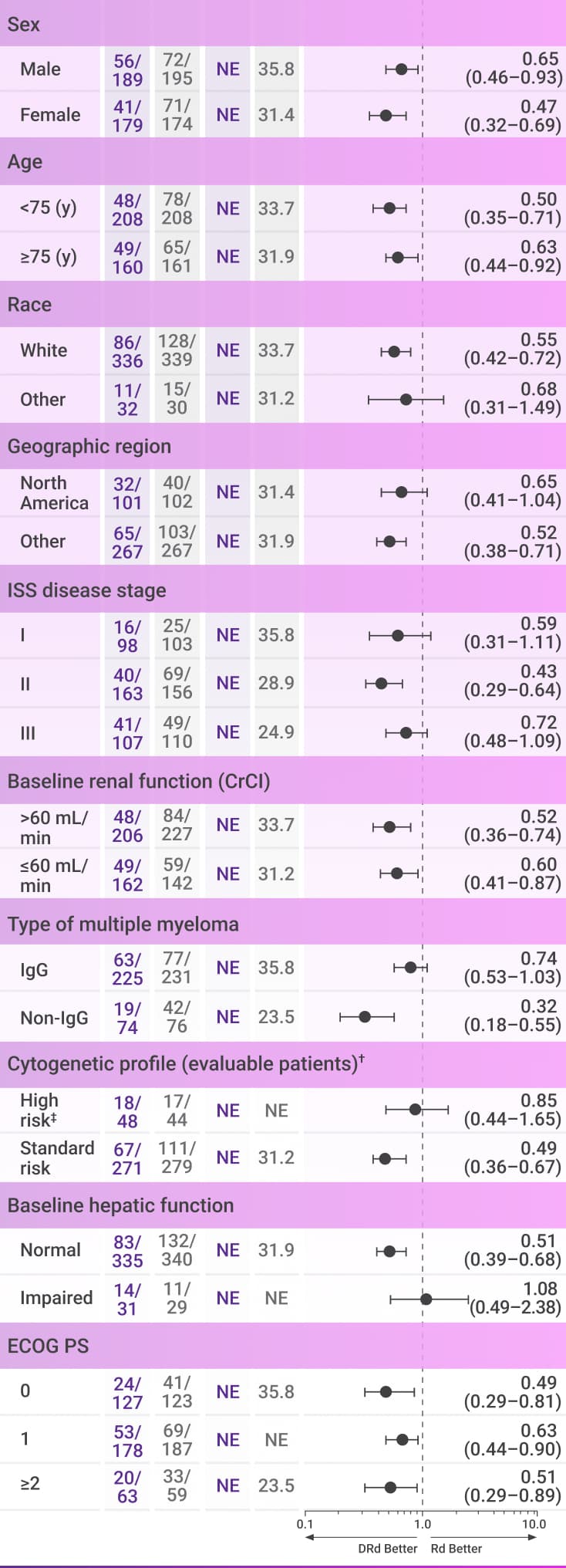
CI=confidence interval; CrCl=creatinine clearance; del=deletion; DRd=DARZALEX® (D) + lenalidomide (R) + dexamethasone (d); ECOG PS=Eastern Cooperative Oncology Group performance status; IgG=immunoglobulin G; ISS=International Staging System; NE=not estimable; PFS=progression-free survival; Rd=lenalidomide (R) + dexamethasone (d); t=translocation.
*Median follow-up was 28 months (range: 0.0-41.4 months).2
†Per protocol, cytogenetic tests from bone marrow aspirates were performed by local laboratories at screening, and 642 patients had baseline cytogenetic data reported from local laboratories out of the 737 patients who were enrolled in MAIA.6,7
‡Cytogenetic risk was based on fluorescence in situ hybridization (FISH) or karyotype analysis; patients who had a high-risk cytogenetic profile had at least one high-risk abnormality (del[17p], t[14;16], or t[4;14]).2
Regardless of age, performance status, or cytogenetic profile:
PFS continued to numerically favor patients on frontline DARZALEX® + Rd vs Rd alone after 56 months of follow-up4*
These analyses are not included in the Prescribing Information for DARZALEX®. These analyses were not adjusted for multiple comparisons. There are insufficient numbers of patients per subgroup to make definitive conclusions of efficacy among the subgroups.
Progression-free survival

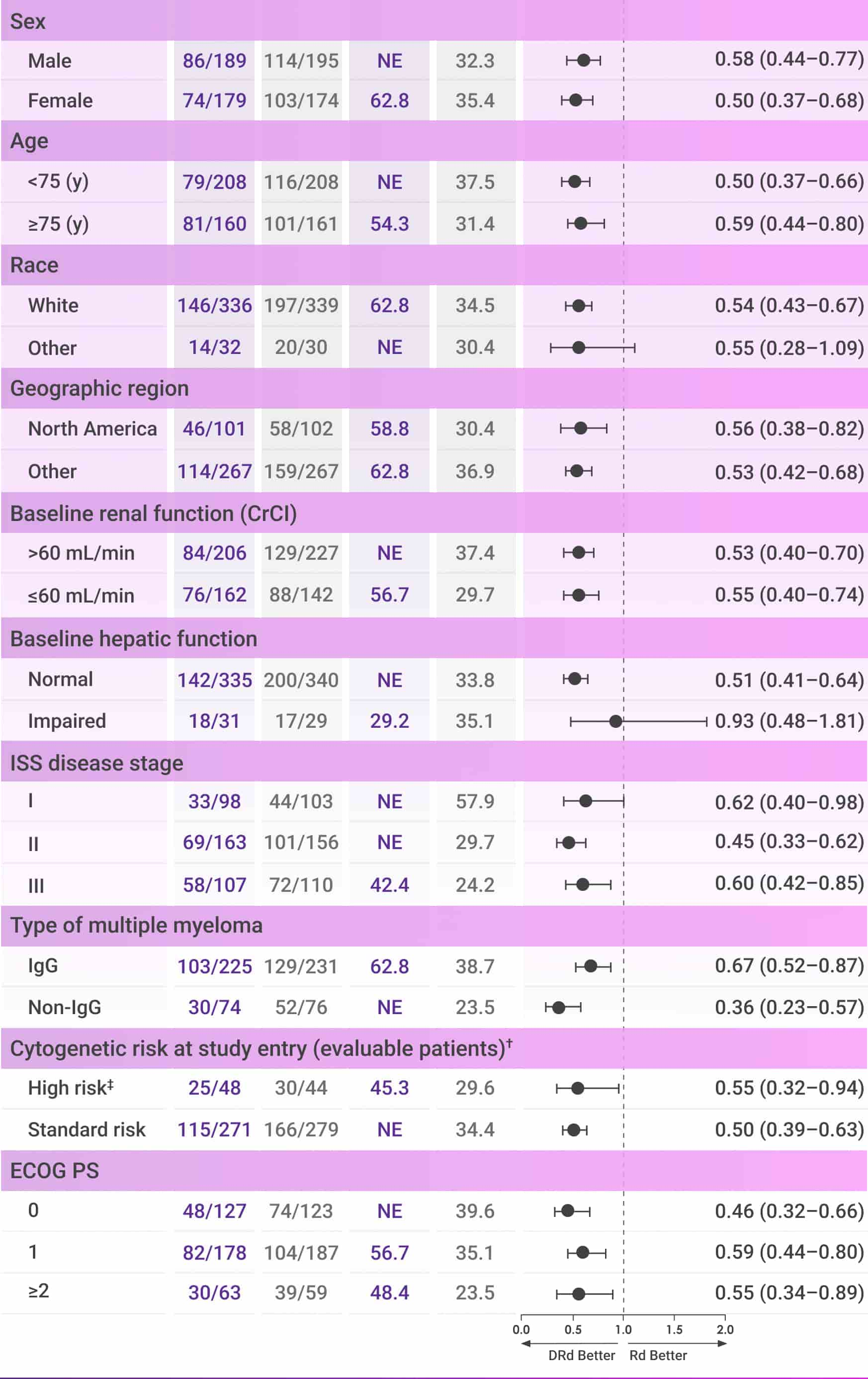

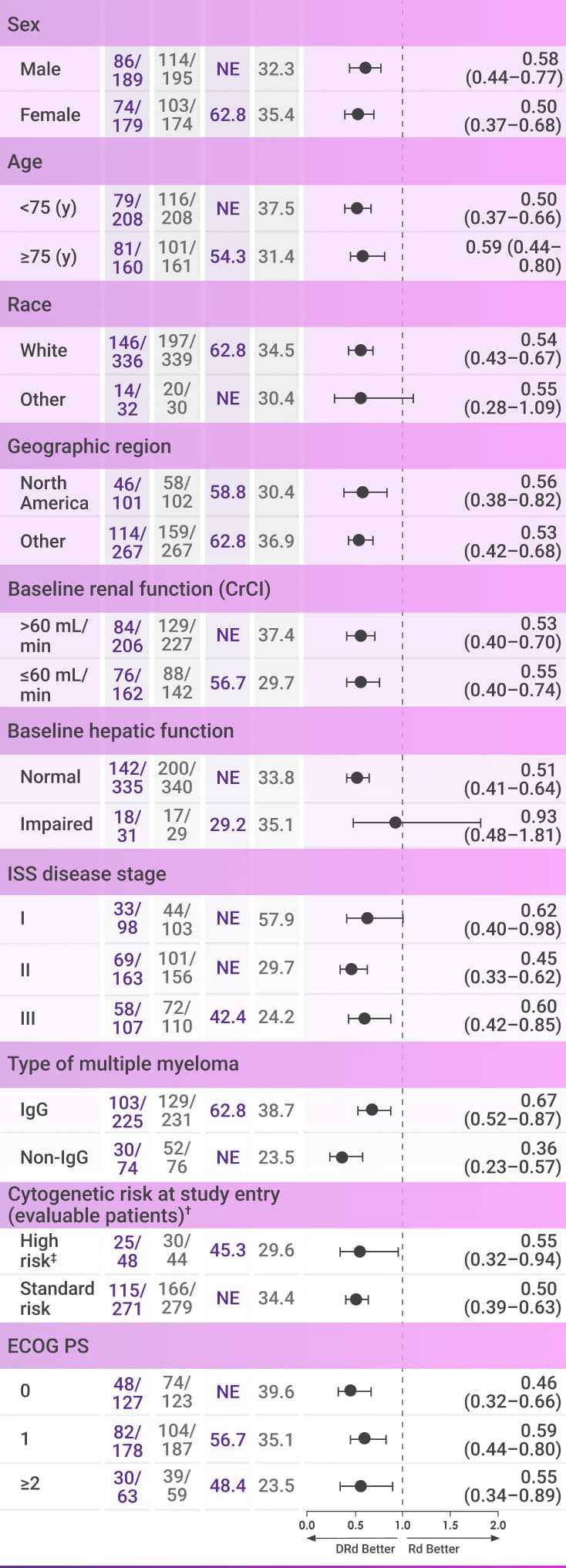
After 56 months of follow-up, the median PFS was not reached with DARZALEX® + Rd vs 34.4 months with Rd alone among the overall MAIA patients (HR=0.53, 95% CI: 0.43-0.66).4*
The follow-up analysis was not adjusted for multiplicity and no conclusions should be drawn.
CI=confidence interval; CrCl=creatinine clearance; DRd=DARZALEX® (D) + lenalidomide (R) + dexamethasone (d); ECOG PS=Eastern Cooperative Oncology Group performance status; IgG=immunoglobulin G; IQR=interquartile range; ISS=International Staging System; NE=not estimable; PFS=progression-free survival; Rd=lenalidomide (R) + dexamethasone (d).
*Median follow-up was 56.6 months in the DRd group (IQR: 53.0–60.1 months) and 55.9 months in the Rd group (IQR: 52.5–59.4 months).4
†Per protocol, cytogenetic tests from bone marrow aspirates were performed by local laboratories at screening and 642 patients had baseline cytogenetic data reported from local laboratories out of the 737 patients who were enrolled in MAIA.6,7
‡Cytogenetic risk was based on fluorescence in situ hybridization (FISH) or karyotype analysis; patients who had a high-risk cytogenetic profile had at least one high-risk abnormality (del[17p], t[14;16], or t[4;14]).2
References:
- DARZALEX® [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc.
- Facon T, Kumar S, Plesner T, et al; the MAIA Trial Investigators. Daratumumab plus lenalidomide and dexamethasone for untreated myeloma. N Engl J Med. 2019;380(22):2104-2115.
- Kumar SK, Moreau P, Bahlis N, et al. Daratumumab plus lenalidomide and dexamethasone (D-Rd) versus lenalidomide and dexamethasone (Rd) alone in transplant-ineligible patients with newly diagnosed multiple myeloma (NDMM): updated analysis of the phase 3 MAIA study. Poster presented at: 64th American Society of Hematology (ASH) Annual Meeting & Exposition; December 10-13, 2022; New Orleans, LA.
- Facon T, Kumar SK, Plesner T, et al. Daratumumab, lenalidomide, and dexamethasone versus lenalidomide and dexamethasone alone in newly diagnosed multiple myeloma (MAIA): overall survival results from a randomised, open-label, phase 3 trial. Lancet Oncol. 2021;22(11):1582-1596.
- Facon T, Kumar SK, Orlowski RZ, et al. Final survival analysis of daratumumab plus lenalidomide and dexamethasone versus lenalidomide and dexamethasone in transplant-ineligible patients with newly diagnosed multiple myeloma: MAIA Study. Presented at: the 2024 European Hematology Association (EHA) Hybrid Congress. Madrid, Spain.
- Moreau P, Facon T, Usmani SZ, et al. Daratumumab plus lenalidomide and dexamethasone (D-Rd) versus lenalidomide and dexamethasone (Rd) in transplant-ineligible patients with newly diagnosed multiple myeloma (NDMM): clinical assessment of key subgroups of the phase 3 MAIA study. Poster presented at: 64th American Society of Hematology (ASH) Annual Meeting & Exposition; December 10-13, 2022; New Orleans, LA.
- Data on file. PM-00913. Janssen Biotech, Inc.



