Frontline DARZALEX® + Rd provides a demonstrated safety profile1*
Discontinuation rates due to any adverse event: 7% with DRd vs 16% with Rd2
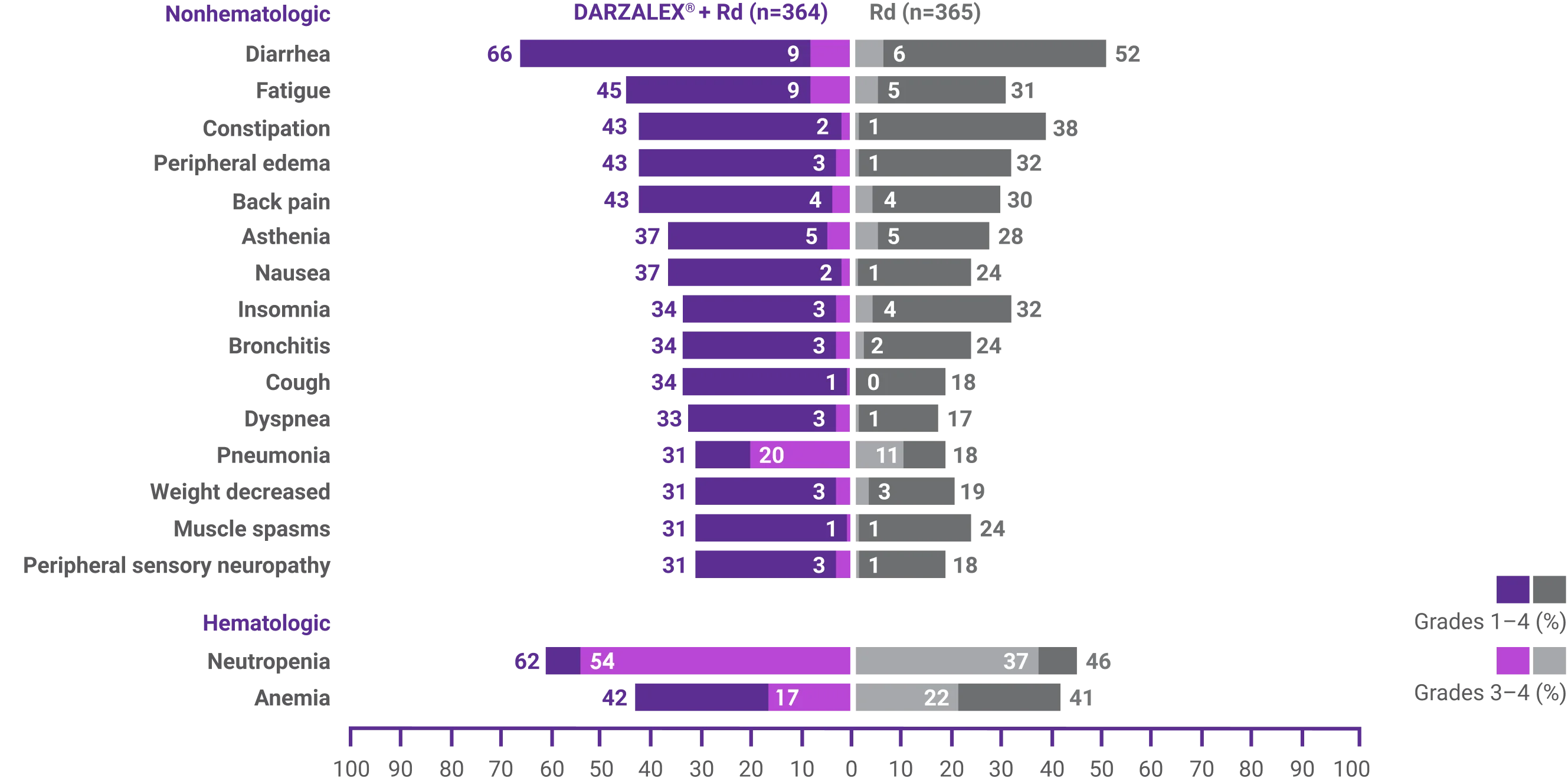
- Infusion-related reactions (IRRs) with DRd occurred in 41% of patients; 2% were Grade 3 and <1% were Grade 41
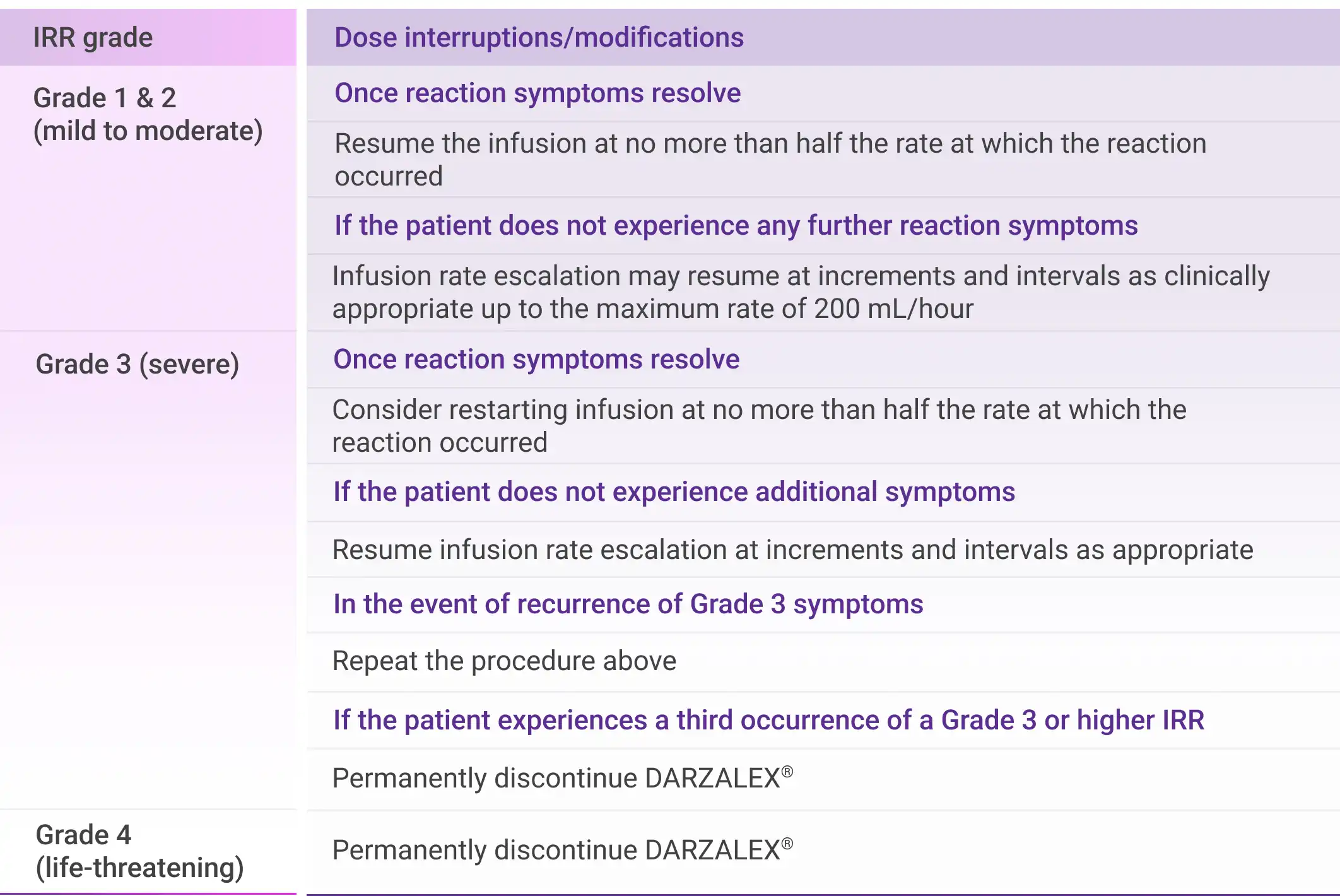
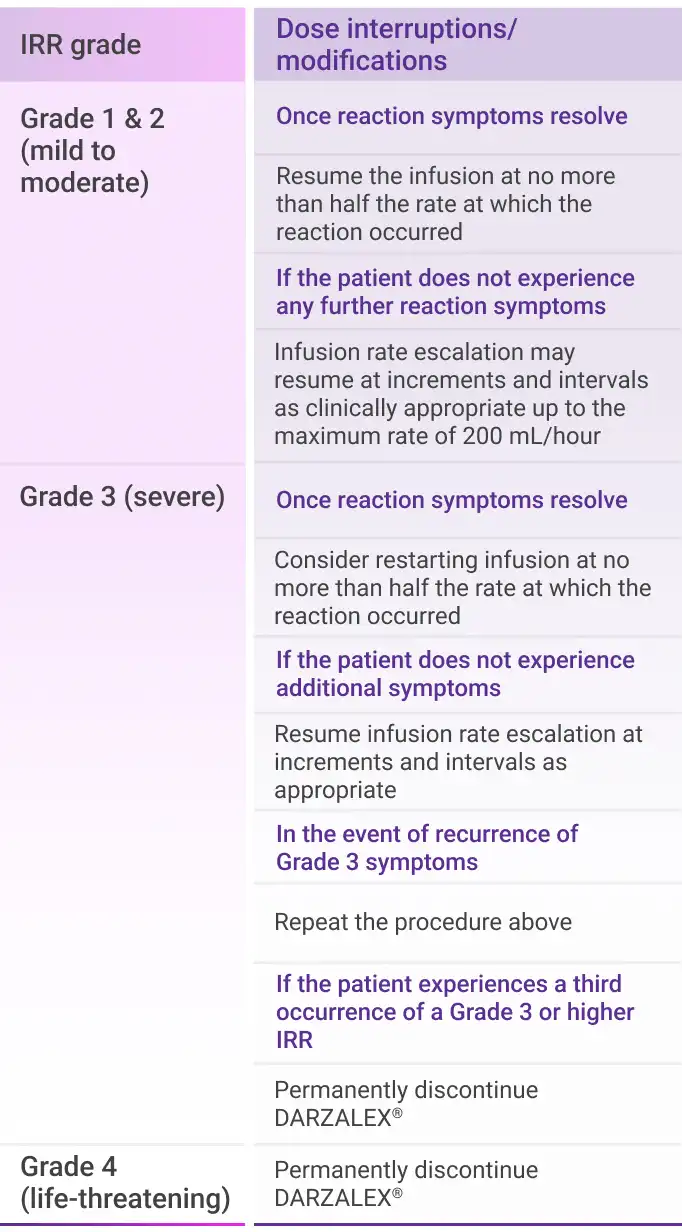
- IRRs of any grade or severity may require management by interruption, modification, and/or discontinuation of the infusion1
- Most IRRs occurred during the first infusion1
DRd=DARZALEX® (D) + lenalidomide (R) + dexamethasone (d); IRR=infusion-related reaction; Rd=lenalidomide (R) + dexamethasone (d)
*Median duration of study treatment was 25.3 months (range: 0.1–40.44 months) in the DRd group and 21.3 months (range: 0.03–40.64 months) in the Rd group.1
†Adverse reactions that occurred with a frequency of ≥10% and <20%, and with at least a 5% greater frequency in DARZALEX® + Rd arm, were headache, urinary tract infection, hyperglycemia, hypocalcemia, vomiting, chills, paresthesia, and hypertension.1
Most frequent adverse reactions reported in ≥10% of patients and with at least 5% greater frequency in DARZALEX® + Rd arm1†
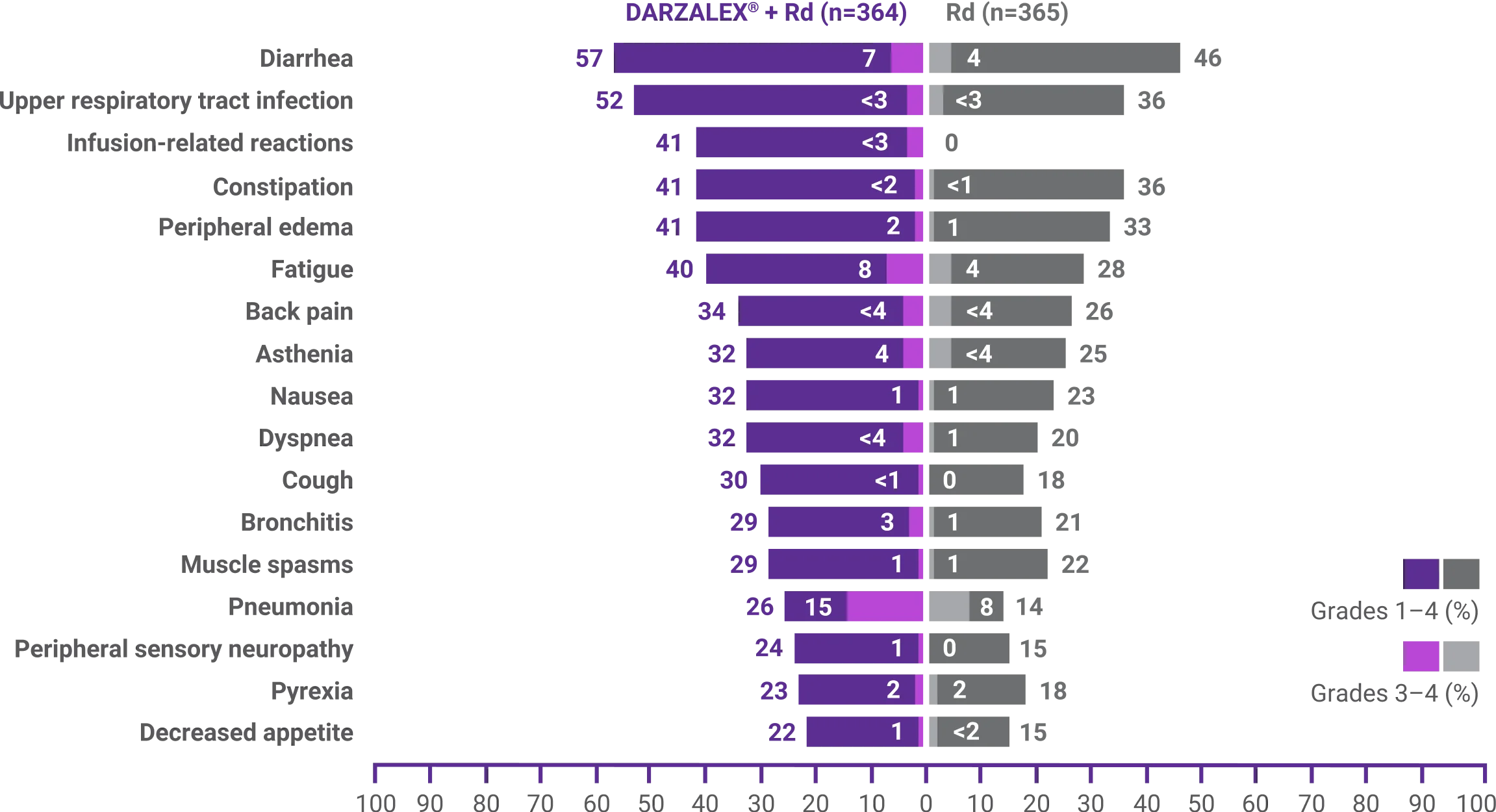

Discontinuation rates due to any adverse event: 7% with DRd vs 16% with Rd2
- Infusion-related reactions (IRRs) with DRd occurred in 41% of patients; 2% were Grade 3 and <1% were Grade 41
- IRRs of any grade or severity may require management by interruption, modification, and/or discontinuation of the infusion1
- Most IRRs occurred during the first infusion1
Most frequent hematologic laboratory abnormalities1
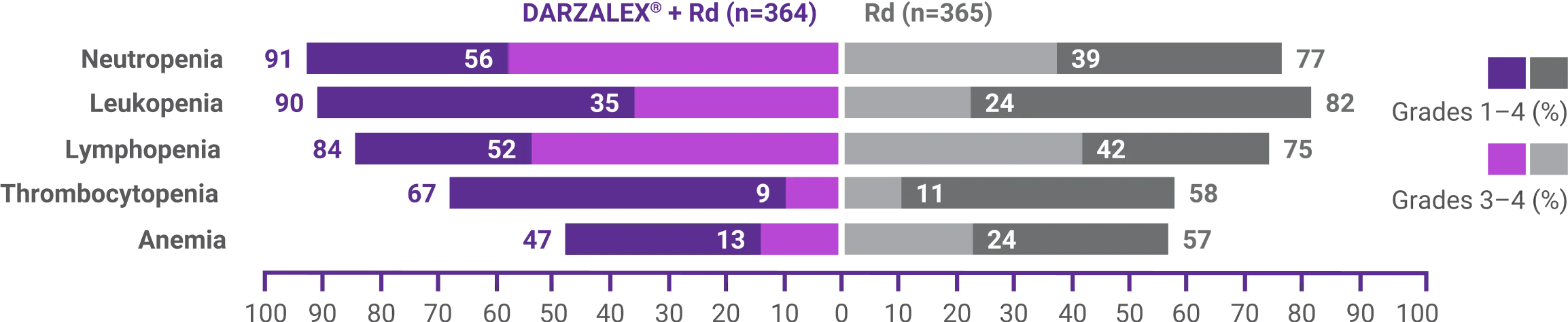

DRd=DARZALEX® (D) + lenalidomide (R) + dexamethasone (d); Rd=lenalidomide (R) + dexamethasone (d)
*Median duration of study treatment was 25.3 months (range: 0.1–40.44 months) in the DRd group and 21.3 months (range: 0.03–40.64 months) in the Rd group.1
†Adverse reactions that occurred with a frequency of ≥10% and <20%, and with at least a 5% greater frequency in DARZALEX® +Rd arm, were headache, urinary tract infection, hyperglycemia, hypocalcemia, vomiting, chills, paresthesia, and hypertension.1
DRd=DARZALEX FASPRO/DARZALEX® (D) + lenalidomide (R) + dexamethasone (d).
First dose of the every-2-week dosing schedule is given at Week 9. First dose of the every-4-week dosing schedule is given at Week 25.1,2
*See the Dosage and Administration section of the full Prescribing Information for more detail. When DARZALEX FASPRO® or DARZALEX® is administered as part of a combination therapy, see the Prescribing Information for dosage recommendations for the other drugs.
†Cycle=28 days.
‡The option of splitting the first dose for DARZALEX® is not applicable to DARZALEX FASPRO®.1,2