MAIA Study Design (DRd)


The MAIA Study: DARZALEX® + Rd
Approval for the treatment of newly diagnosed, transplant-ineligible patients with multiple myeloma was based on a large, randomized, open-label, multicenter, active-controlled, phase 3 trial.1,2
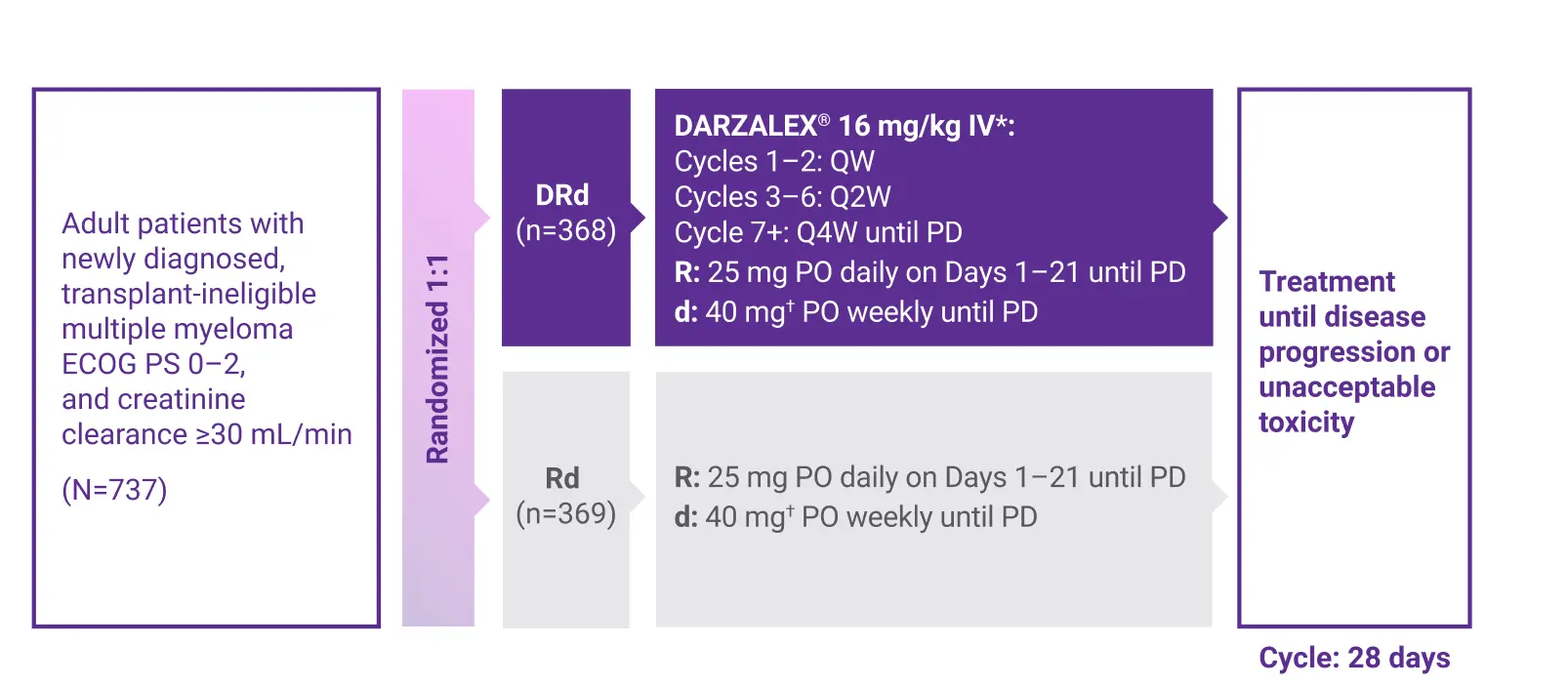

Primary endpoint:
Progression-free survival (PFS) based on International Myeloma Working Group (IMWG) criteria2
Key secondary endpoints included:
Percentage of patients with complete response (CR), very good partial response (VGPR) rate, minimal residual disease (MRD) negativity rate (next-generation sequencing [NGS]; 10–5), overall response rate (ORR), overall survival (OS), duration of response, and safety1,2
Baseline demographics and disease characteristics:
Were similar between the 2 treatment groups1
d=dexamethasone; DRd=DARZALEX® (D) + lenalidomide (R) + dexamethasone (d); ECOG PS=Eastern Cooperative Oncology Group performance status; IV=intravenous; PD=progressive disease; PO=by mouth; QW=weekly; Q2W=every 2 weeks; Q4W=every 4 weeks; R=lenalidomide; Rd=lenalidomide (R) + dexamethasone (d).
*On days when daratumumab was administered, dexamethasone was administered to patients in the DRd arm and served as the treatment dose of steroid for that day, as well as the required pre-infusion medication.1
†For patients older than 75 years of age or with body mass index (BMI) <18.5, dexamethasone was administered at a dose of 20 mg weekly.1
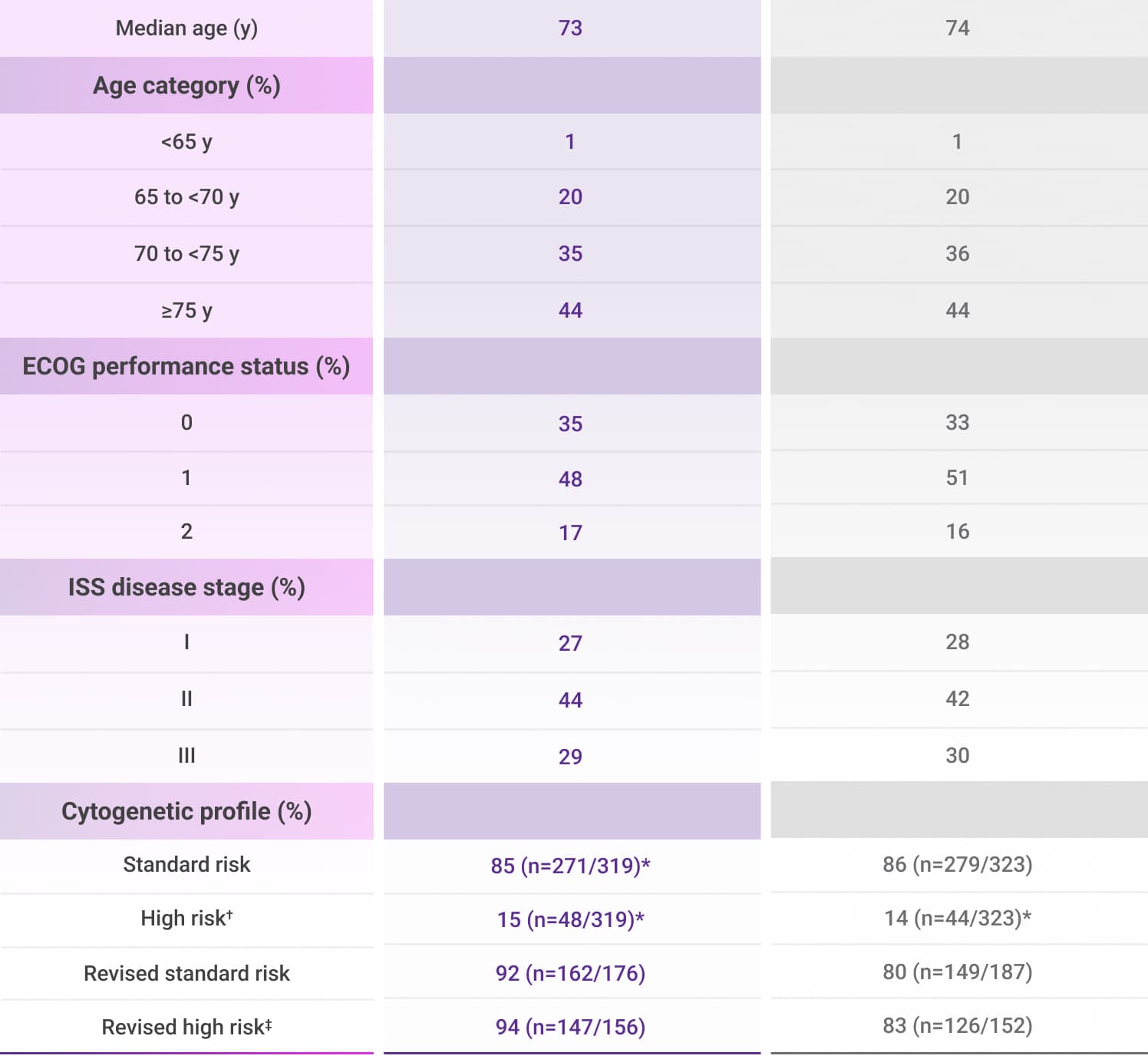
MAIA Patient Demographics
Frontline DARZALEX® + Rd—evaluated in a wide range of newly diagnosed, transplant-ineligible patients2-4
The MAIA trial included patients of various ages, ECOG PS, and prognostic factors2
- Approximately half (44%) of patients were 75 years of age or older2
amp=amplification; del=deletion; ECOG PS=Eastern Cooperative Oncology Group performance status; ISS=International Staging System; Rd=lenalidomide (R) + dexamethasone (d); t=translocation.
*Per protocol, cytogenetic tests from bone marrow aspirates were performed by local laboratories at screening, and 642 patients had baseline cytogenetic data reported from local laboratories out of the 737 patients who were enrolled in the MAIA trial.5,6
†Cytogenetic risk was based on fluorescence in situ hybridization (FISH) or karyotype analysis; patients who had a high-risk cytogenetic profile had at least one high-risk abnormality (del[17p], t[14;16], or t[4;14]).2
‡Revised high-risk cytogenetics is defined as having at least 1 out of the following 6 abnormalities: t(4;14), t(14;16), del(17p), t(14;20), gain(1q21), or amp(1q21).4


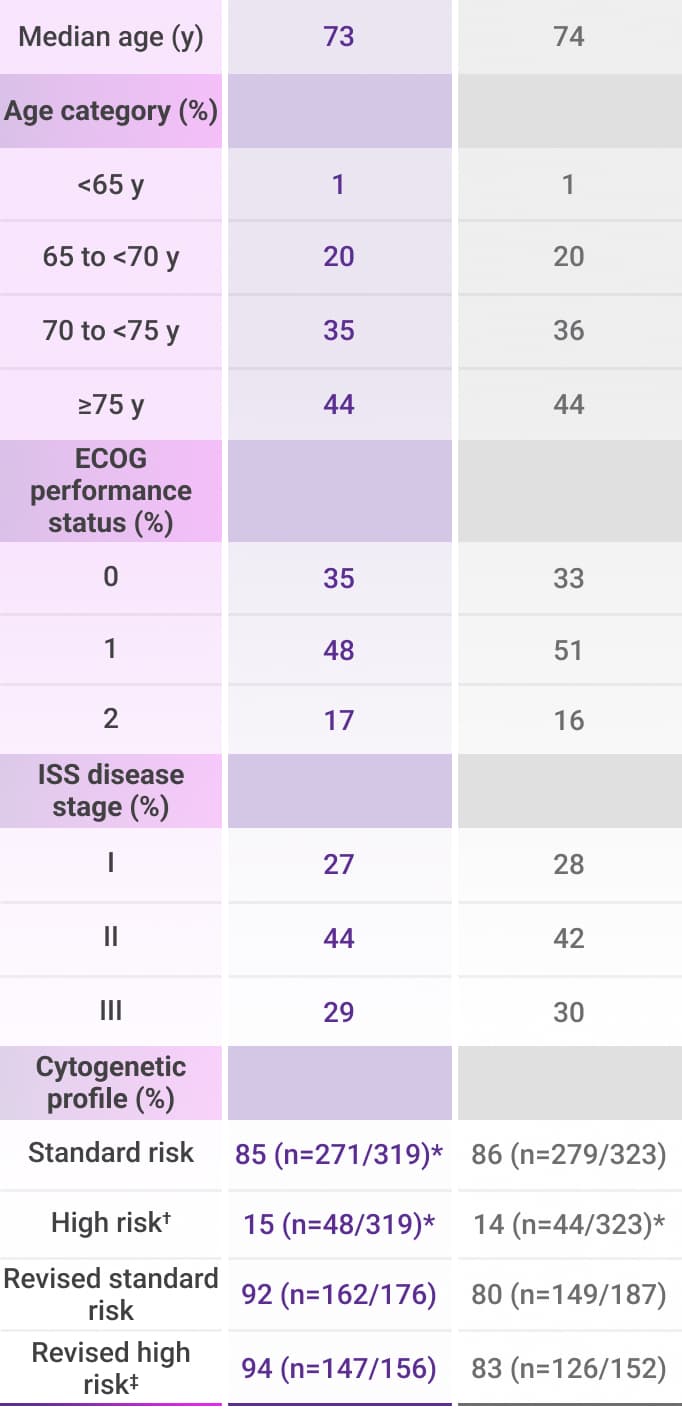
amp=amplification; del=deletion; ECOG PS=Eastern Cooperative Oncology Group performance status; ISS=International Staging System; Rd=lenalidomide (R) + dexamethasone (d).
*Per protocol, cytogenetic tests from bone marrow aspirates were performed by local laboratories at screening and 642 patients had baseline cytogenetic data reported from local laboratories out of the 737 patients who were enrolled in the MAIA trial.5,6
†Cytogenetic risk was based on fluorescence in situ hybridization (FISH) or karyotype analysis; patients who had a high-risk cytogenetic profile had at least one high-risk abnormality (del[17p], t[14;16], or t[4;14]).2
Transplant-Ineligible Multiple Myeloma
NCCN Category 1, preferred*
The NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) may contain data, conclusions, and recommendations that do not conform to the FDA-approved labeling for the products discussed herein and have not been evaluated by the FDA. No conclusions should be drawn. Daratumumab (DARZALEX®) and daratumumab and hyaluronidase-fihj (DARZALEX FASPRO®) should be used only as specified in the Prescribing Information.
Daratumumab† (D) in combination with lenalidomide (R) and dexamethasone (d) is recommended by the NCCN Guidelines® as a Category 1 preferred therapeutic option for patients with newly diagnosed multiple myeloma when hematopoietic stem cell transplant is deferred or not indicated.7
Safety and effectiveness of DRd in the transplant-deferred patient population has not been evaluated in a phase 3 study and has not been established.
FDA-Approved Indication
Daratumumab (DARZALEX®) and daratumumab and hyaluronidase-fihj (DARZALEX FASPRO®) are indicated for the treatment of adult patients with multiple myeloma in combination with lenalidomide and dexamethasone in newly diagnosed patients who are ineligible for autologous stem cell transplant.1,8
*See NCCN.org for definitions of NCCN Categories of Preference and NCCN Categories of Evidence and Consensus.
†Daratumumab includes both daratumumab (DARZALEX®) for intravenous infusion and daratumumab and hyaluronidase-fihj (DARZALEX FASPRO®) for subcutaneous injection. Daratumumab and hyaluronidase-fihj for subcutaneous injection has different dosing and administration instructions compared with daratumumab for intravenous infusion.
References:
DARZALEX FASPRO® [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc.
Facon T, Kumar S, Plesner T, et al; the MAIA Trial Investigators. Daratumumab plus lenalidomide and dexamethasone for untreated myeloma. N Engl J Med. 2019;380(22):2104-2115.
Facon T, Kumar SK, Plesner T, et al. Daratumumab, lenalidomide, and dexamethasone versus lenalidomide and dexamethasone alone in newly diagnosed multiple myeloma (MAIA): overall survival results from a randomised, open-label, phase 3 trial. Lancet Oncol. 2021;22(11):1582-1596.
Moreau P, Facon T, Usmani SZ, et al. Daratumumab plus lenalidomide/dexamethasone in untreated multiple myeloma: analysis of key subgroups of the MAIA study. Leukemia. 2025;39:710-719.
Moreau P, Facon T, Usmani SZ, et al. Daratumumab plus lenalidomide and dexamethasone (D-Rd) versus lenalidomide and dexamethasone (Rd) in transplant-ineligible patients with newly diagnosed multiple myeloma (NDMM): clinical assessment of key subgroups of the phase 3 MAIA study. Poster presented at: 64th American Society of Hematology (ASH) Annual Meeting & Exposition; December 10-13, 2022; New Orleans, LA.
Data on file. PM-00913. Janssen Biotech, Inc.
Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Multiple Myeloma V.5.2026. © National Comprehensive Cancer Network, Inc. 2026. All rights reserved. Accessed January 12, 2026. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way.
- DARZALEX® [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc.

