Frailty Subgroup
(Post Hoc Analysis)


Examine Samuel’s case for a deeper understanding of patients for whom frontline DRd may be appropriate.
Meet Samuel (Age 78): Elderly patient with comorbidities and identified as frail by his primary care provider*


*Hypothetical patient case.
Newly diagnosed and not
eligible for transplant
- History of working in construction but now retired
- Good support system: not married but in a steady relationship past 20 years
- At times, struggles to stay adherent to treatment
Clinical condition and disease presentation
Additional considerations
- Manages diabetes with regular glucose monitoring, insulin, and coordinated care with his healthcare team
- Requires assistance to get around, especially up and down stairs
Transplant eligibility of an individual patient is determined based on evaluation by the treating physician.
DRd=DARZALEX® (D) + lenalidomide (R) + dexamethasone (d); ECOG PS=Eastern Cooperative Oncology Group performance status; ISS=International Staging System.
This analysis is not included in the Prescribing Information for DARZALEX®. This analysis was conducted post hoc, and there are insufficient numbers of patients per subgroup to make definitive conclusions of efficacy among the subgroups.
Please see MAIA trial design & primary results
Post hoc subgroup analysis of MAIA by frailty status score
- The median age in frail subgroup was 77 years (range: 57-90 years), with 88% of patients having ECOG PS ≥11
- CCI was calculated based on retrospective review of each patient’s medical history1
- Frailty assessment was performed retrospectively using age, CCI, and baseline ECOG PS1
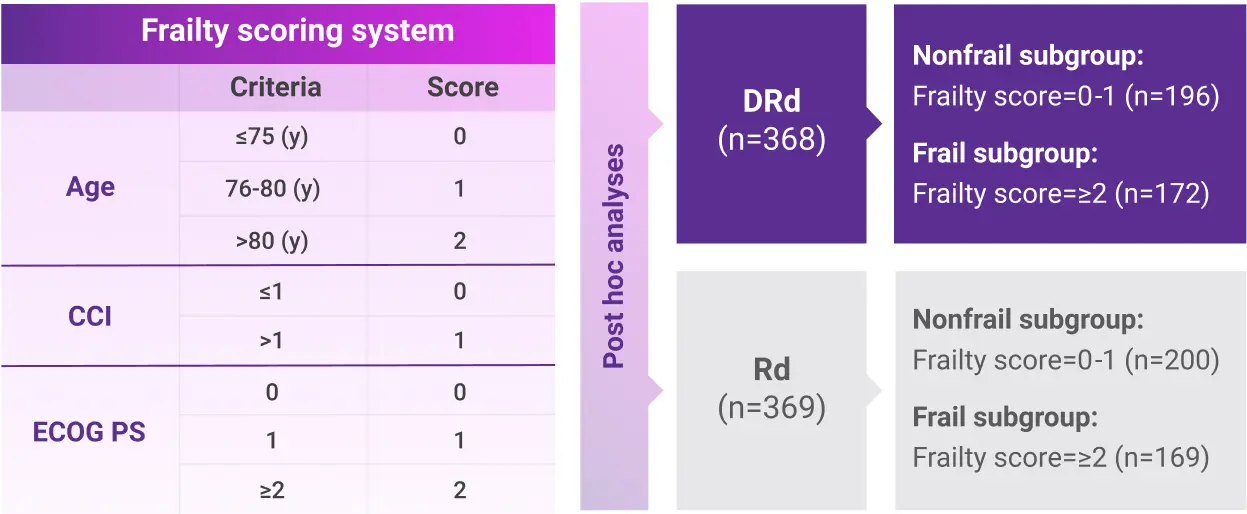

- The median age in frail subgroup was 77 years (range: 57-90 years), with 88% of patients having ECOG PS ≥13
- CCI was calculated based on retrospective review of each patient’s medical history3
- Frailty assessment was performed retrospectively using age, CCI, and baseline ECOG PS3
The retrospective assessment of frailty score was a limitation of this study. Retrospective CCI calculations were based on reported medical history, which may contain missing data and result in underestimating or overestimating the number of patients in each frailty subgroup. The ECOG PS score parameter used for frailty score calculations in the study is more subjective, with susceptibility to intra- and inter-observer bias, compared with the ADL and IADL scales used in the IMWG scoring system. While the frailty scale used in the study is based on parameters that are routinely assessed in clinical practice for clinical use, the use of comprehensive frailty assessments that more accurately reflect biological or functional frailty will remain important for the further optimization of treatment strategies for frail patients. Patients with an ECOG PS score ≥3 and patients with comorbidities that may interfere with the study procedures were excluded from MAIA; the inclusion and exclusion criteria for the study limit the generalizability of these results to more frail patients seen in clinical practice.1
ADL=activities of daily living; CCI=Charlson Comorbidity Index; DRd=DARZALEX® (D) + lenalidomide (R) + dexamethasone (d); ECOG PS=Eastern Cooperative Oncology Group performance status; IADL=instrumental activities of daily living; IMWG=International Myeloma Working Group; Rd=lenalidomide (R) + dexamethasone (d).
In a subgroup analysis of frail patients after 36.4 months of follow-up:
Progression-Free Survival (PFS)1
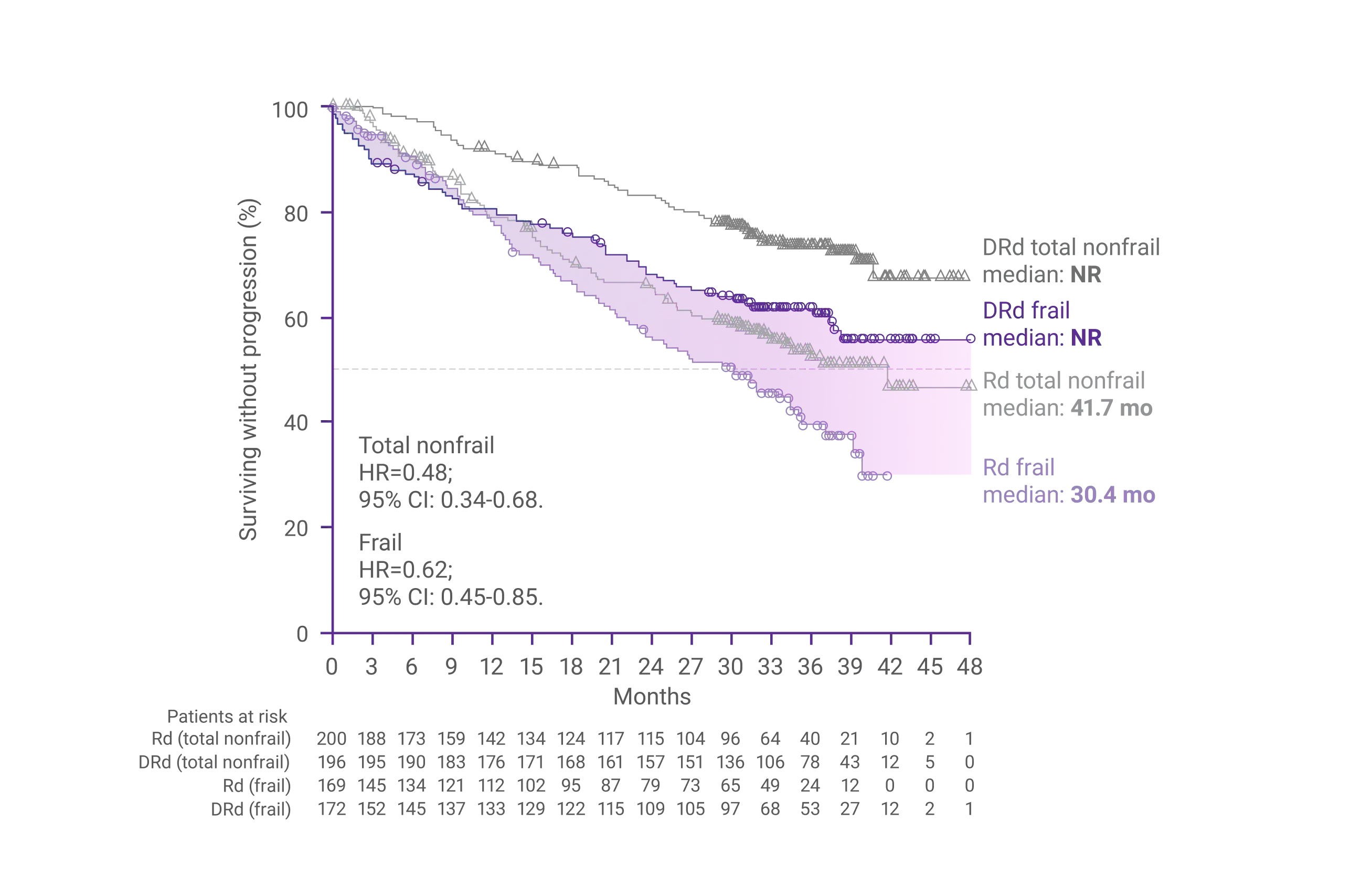
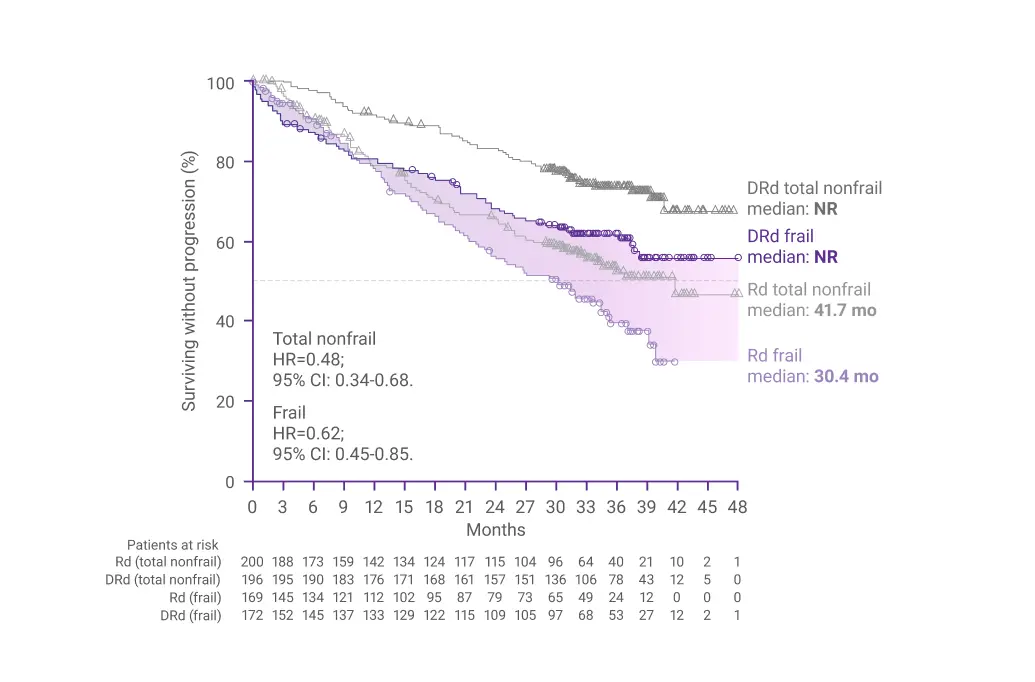
In frail patients, the risk reduction for disease progression or death was 38% with DRd vs Rd alone (HR=0.62; 95% CI: 0.45-0.85)1
CI=confidence interval; DRd=DARZALEX® (D) + lenalidomide (R) + dexamethasone (d); HR=hazard ratio; NR=not reached; PFS=progression-free survival; Rd=lenalidomide (R) + dexamethasone (d).
*Median follow-up was 36.4 months.1
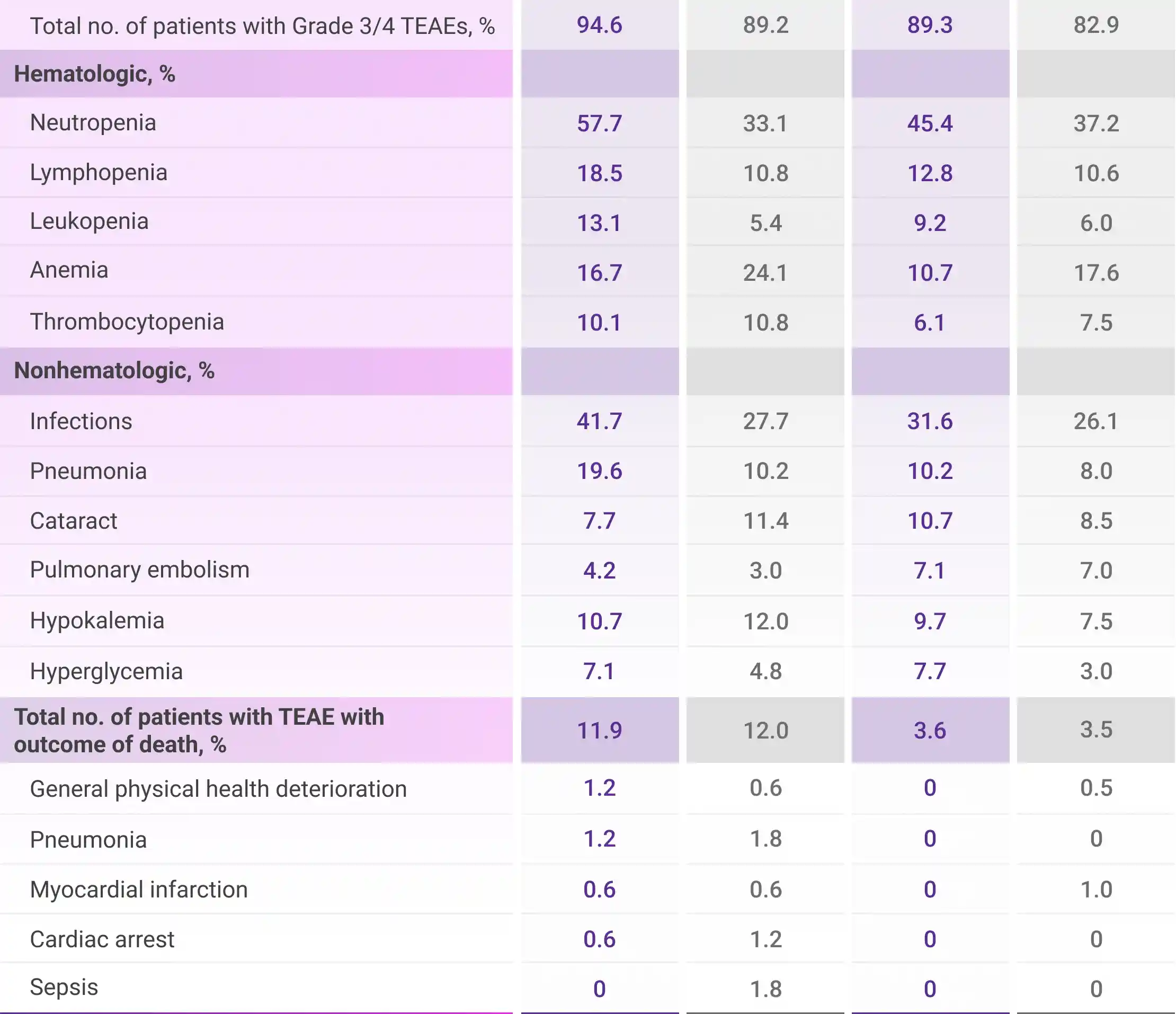
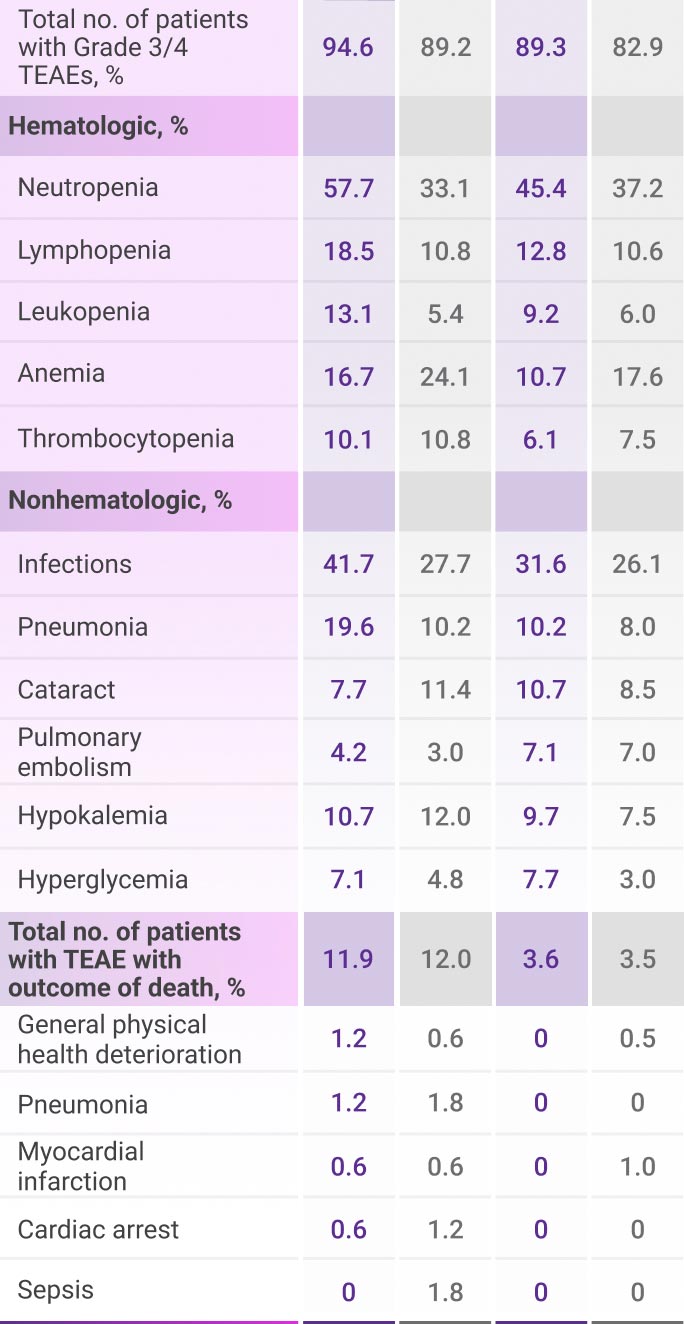
Most frequent Grade 3/4 TEAEs (≥10%) in frail patients1
All TEAEs are reported as observed. This analysis was conducted post hoc and no conclusions should be drawn.
DRd=DARZALEX® (D) + lenalidomide (R) + dexamethasone (d); Rd=lenalidomide (R) + dexamethasone (d); TEAE=treatment-emergent adverse event.


Of the 2,459 patients who received DARZALEX® at the recommended dose, 38% were 65 to 74 years of age, and 15% were 75 years of age or older. No overall differences in effectiveness were observed between these patients and younger patients. The incidence of serious adverse reactions was higher in older patients than in younger patients. Among patients with relapsed and refractory multiple myeloma (n=1,213), the serious adverse reactions that occurred more frequently in patients 65 years and older were pneumonia and sepsis. Among patients with newly diagnosed multiple myeloma who were ineligible for autologous stem cell transplant (n=710), the serious adverse reaction that occurred more frequently in patients 75 years and older was pneumonia.2
Of the 214 patients who received DARZALEX FASPRO® as combination therapy with pomalidomide and dexamethasone or DARZALEX FASPRO® as combination therapy with lenalidomide and low-dose dexamethasone for relapsed and refractory multiple myeloma, 43% were 65 to <75 years of age, and 18% were 75 years of age or older.3
Adverse reactions occurring at a higher frequency (≥5% difference) in patients ≥65 years of age included fatigue, pyrexia, peripheral edema, urinary tract infection, diarrhea, constipation, vomiting, dyspnea, cough, and hyperglycemia. Serious adverse reactions occurring at a higher frequency (≥2% difference) in patients ≥65 years of age included neutropenia, thrombocytopenia, diarrhea, anemia, COVID-19, ischemic colitis, deep vein thrombosis, general physical health deterioration, pulmonary embolism, and urinary tract infection.3
No clinically meaningful differences in the pharmacokinetics of daratumumab were observed in geriatric patients compared to younger adult patients.3
DRd=DARZALEX® (D) + lenalidomide (R) + dexamethasone (d); Rd=lenalidomide (R) + dexamethasone (d); TEAE=treatment-emergent adverse event.
References:
- Facon T, Cook G, Usmani SZ, et al. Daratumumab plus lenalidomide and dexamethasone in transplant-ineligible newly diagnosed multiple myeloma: frailty subgroup analysis of MAIA. Leukemia. 2022;36(4):1066-1077.
- DARZALEX® [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc.
- DARZALEX FASPRO® [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc.

